
The H5N1 Spillover In United States Of America: Was It Preventable?
The H5N1 Spillover signal was not a siren, but a silence in the milking parlors of the Texas Panhandle. In early February 2024, dairy producers in Hartley, Moore, and Castro counties began reporting a “mystery illness” that standard veterinary diagnosis could not detect. High-yielding Holsteins suddenly grew lethargic, their feed intake plummeted, and their milk, a pristine white fluid, thickened into a colostrum-like sludge. For nearly six weeks, this biological anomaly went unclassified, allowing the H5N1 virus (Clade 2. 3. 4. 4b, Genotype B3. 13) to circulate unchecked through the nation’s most concentrated dairy corridor before federal regulators identified the pathogen on March 25, 2024.
The timeline of this H5N1 spillover reveals a serious gap between the biological onset of the outbreak and the regulatory response. While the United States Department of Agriculture (USDA) confirmed the presence of Highly Pathogenic Avian Influenza (HPAI) in late March, retrospective analysis places the index infection events as early as late January or early February. During this roughly 60-day blind spot, the virus utilized cattle transport networks to seed infections in Kansas, Michigan, and New Mexico, outmaneuvering containment before they were even drafted.
The “Mystery Illness” Phase: February 1 , March 20
By mid-February, the Texas Association of Dairymen was fielding reports of a contagion moving “herd to herd.” The clinical presentation was distinct: older cows in mid-lactation were hit hardest, with milk production dropping by 10% to 20% across affected herds. Unlike respiratory flu presentations in swine or humans, the primary vector of viral shedding in these cattle was mammary. The milk itself carried viral loads exceeding those found in the respiratory tracts of infected poultry, turning the milking into a high-efficiency transmission vector.
The diagnostic delay stemmed from a rigid biological assumption: cows do not get Influenza A. Consequently, samples sent to the Texas A&M Veterinary Medical Diagnostic Laboratory (TVMDL) were initially screened for common bovine pathogens, bovine viral diarrhea, pneumonia, and mastitis. All returned negative. It was only after significant mortality events among farm cats, who drank raw colostrum and subsequently died with neurological symptoms, and the discovery of dead grackles and blackbirds around the dairies, that the testing parameters were widened to include avian influenza.
| Date Range | Event Description | Key Metrics / Observations |
|---|---|---|
| Feb 5 , Feb 15 | Initial reports of “mystery illness” in Panhandle herds. | Milk production drops 10-20%; thick, discolored milk reported. |
| Feb 20 , Mar 10 | Spread to additional herds; interstate cattle movements continue. | Reports of dead barn cats and wild birds on premises. |
| Mar 15 , Mar 19 | TVMDL receives samples specifically for broad viral testing. | Testing expands beyond standard bovine panels. |
| Mar 21 , Mar 22 | Samples from Kansas and Texas test non-negative for Influenza A. | indication of zoonotic spillover to bovine hosts. |
| Mar 25 | USDA / APHIS confirms H5N1 in Texas and Kansas dairy cattle. | Official confirmation date. US detection in cattle. |
| Apr 1 | human case confirmed in Texas dairy worker. | Patient presents with conjunctivitis; history of direct cow exposure. |
The Human Index Case: April 1, 2024
The spillover event culminated in the confirmed human infection linked to the dairy reservoir, reported by the Texas Department of State Health Services on April 1, 2024. The patient, a dairy worker in the Panhandle region, presented with conjunctivitis (pink eye) rather than severe respiratory distress. This clinical manifestation aligned with the high viral concentrations found in the milk; direct contact with the eyes via contaminated gloves or equipment was the likely route of transmission. While the worker recovered, the case shattered the species barrier assumption that had previously governed H5N1 risk assessments for livestock workers.
The delay in identifying the cattle reservoir meant that for nearly two months, farm workers were exposed to high-titer virus without enhanced personal protective equipment (PPE). The Centers for Disease Control and Prevention (CDC) subsequently issued alerts, yet the window for early containment had closed. Genomic sequencing later revealed that the virus had already undergone reassortment, acquiring adaptations that facilitated its persistence in the bovine mammary gland. The failure to test for influenza in cattle during February 2024 stands as the primary procedural error that transformed a local spillover into a multi-state agricultural emergency.
Genomic Forensics: Tracing the B3. 13 Genotype Origin
The viral agent responsible for the Texas panhandle outbreak was not a generic avian influenza a specific, highly adapted biological entity: H5N1 clade 2. 3. 4. 4b, genotype B3. 13. Genomic sequencing conducted by the USDA and academic laboratories confirms that this genotype emerged from a precise reassortment event, a genetic shuffling, between the Eurasian panzootic H5N1 virus and low-pathogenicity avian influenza (LPAI) viruses circulating in North American wild birds. This fusion likely occurred in late 2023, creating a chimeric pathogen capable of breaching the bovine species barrier.
Phylogenetic analysis reveals that B3. 13 is a “4+4” reassortant. It retains four gene segments from the highly pathogenic Eurasian lineage (Hemagglutinin [HA], Neuraminidase [NA], Matrix [MP], and Polymerase Acidic [PA]) and acquired four internal gene segments from North American wild bird lineages (Polymerase Basic 2 [PB2], Polymerase Basic 1 [PB1], Nucleoprotein [NP], and Non-structural [NS]). This specific combination provided the virus with the external to enter cells and the internal replication engines adapted to the local host environment.
The forensic timeline places the spillover event well before the public confirmations in March 2024. Molecular clock dating, which estimates the time to the Most Recent Common Ancestor (tMRCA), indicates that the B3. 13 genotype likely jumped from a wild bird reservoir to cattle between November 2023 and January 2024. The data suggests a single, point-source introduction in the Texas Panhandle, followed by rapid lateral transmission among herds. This “single spillover” theory contradicts early assumptions of multiple, independent introductions from wild birds, pointing instead to sustained cow-to-cow transmission as the primary driver of the epizootic.
A central factor in this successful host jump was a specific mutation in the viral polymerase: PB2 M631L. This amino acid substitution, methionine replaced by leucine at position 631 of the PB2 protein, was present in all sequenced cattle samples. Functional assays demonstrate that the M631L mutation allows the avian viral polymerase to hijack the bovine ANP32A protein, a cellular cofactor necessary for viral replication. Without this molecular key, the avian virus would have struggled to replicate within bovine cells. The ubiquity of M631L in the cattle clade indicates it was either a prerequisite for the initial spillover or was selected for immediately upon entry into the bovine population.
The genomic evidence also exposes the extent of silent circulation. By the time federal regulators announced the outbreak on March 25, 2024, the virus had already established a distinct monophyletic clade. The genetic diversity observed within this clade implies months of transmission, allowing the virus to refine its replication strategies in the new host. Later analysis identified additional polymerase mutations, such as PB2 Q591R and PA K497R, in specific clusters, signaling ongoing adaptation as the virus moved through the U. S. dairy herd.
Table: Genomic Composition of H5N1 Genotype B3. 13
The following table details the lineage origins of the eight gene segments comprising the B3. 13 genotype found in the Texas cattle outbreak.
| Gene Segment | Function | Lineage Origin | Role in Pathogenesis |
|---|---|---|---|
| HA (Hemagglutinin) | Viral entry / Receptor binding | Eurasian H5N1 (2. 3. 4. 4b) | Retains high pathogenicity; receptors. |
| NA (Neuraminidase) | Viral release | Eurasian H5N1 (2. 3. 4. 4b) | spread between cells. |
| PB2 (Polymerase Basic 2) | Replication / Host adaptation | North American Wild Bird (LPAI) | Carries M631L mutation for bovine replication. |
| PB1 (Polymerase Basic 1) | RNA synthesis | North American Wild Bird (LPAI) | Supports viral genome replication. |
| PA (Polymerase Acidic) | Endonuclease activity | Eurasian H5N1 (2. 3. 4. 4b) | Works with PB2/PB1 for replication. |
| NP (Nucleoprotein) | RNA packaging | North American Wild Bird (LPAI) | Encapsidates viral RNA. |
| MP (Matrix) | Structural integrity | Eurasian H5N1 (2. 3. 4. 4b) | Maintains virion stability. |
| NS (Non-structural) | Immune evasion | North American Wild Bird (LPAI) | Antagonizes host interferon response. |
The emergence of genotype B3. 13 represents a significant biological shift. While previous H5N1 incursions into mammals were frequently dead-end spillover events, the B3. 13 genotype demonstrated the capacity for sustained transmission within a mammalian reservoir. The acquisition of the North American PB2 segment, followed by the M631L adaptation, transformed a bird virus into a pathogen capable of exploiting the industrial dairy environment. This genomic reality challenges the initial regulatory narrative of a contained, minor event and confirms that the biological for a mammalian epizootic was fully assembled months before the official response began.
The Migratory Vector: Wild Bird Interface Failures
The biological between the avian reservoir and the Texas dairy herd was not built on a single day, through months of unchecked interface at the agricultural margins. Throughout late 2023 and early 2024, the Central and Mississippi flyways funneled millions of migratory waterfowl directly over the Texas Panhandle, a region that concentrates over 400, 000 dairy cows in open-lot systems. This convergence created a high-probability zone for spillover that federal surveillance systems, designed primarily for poultry defense, failed to flag for the dairy sector.
Genomic analysis of the outbreak, identified as H5N1 genotype B3. 13, confirms its origin in a reassortment event between Eurasian clade 2. 3. 4. 4b viruses and low-pathogenicity North American avian flu. This specific genotype emerged in wild bird populations in late 2023, carried by mallards and other dabbling ducks that frequent agricultural wastewater lagoons. Unlike poultry operations, which are frequently enclosed and biosecure, Panhandle dairy farms use open-air feed bunks and massive water troughs. These features acted as biological attractants for synanthropic species, grackles, European starlings, and rock pigeons, that the gap between migratory waterfowl and livestock.
Field reports from January and February 2024 document a series of missed ecological warning signs. Veterinary logs from Hartley and Castro counties note an unusual uptick in “perimeter mortality” involving grackles and blackbirds found dead near cattle feed lanes. These incidents coincided with the initial drops in milk production were not immediately tested for H5N1. The failure to link these wild bird die-offs to the emerging bovine syndrome represents a serious lapse in One Health surveillance. State diagnostic data reveals that while poultry flocks were under strict monitoring, dairy biosecurity regarding wild birds were virtually nonexistent, with no federal guidance issued to dairy producers regarding waterfowl risks prior to March 2024.
| Interface Point | Risk method | Observed Vector Species | Regulatory Status (Pre-March 2024) |
|---|---|---|---|
| Open Feed Bunks | Fecal contamination of Total Mixed Ration (TMR) | Great-tailed Grackles, Starlings | Unregulated / No Exclusion Required |
| Wastewater Lagoons | Viral persistence in water; waterfowl congregation | Mallards, Northern Pintails | Standard EPA/TCEQ compliance only |
| Water Troughs | Direct oral-fecal transmission via shared drinking | Rock Pigeons, Sparrows | No biosecurity separation mandates |
| Silage Piles | Rodent and bird nesting in stored feed | House Mice, Starlings | General sanitation guidelines only |
The transmission mechanics were brutally. Investigations by the Texas A&M Veterinary Medical Diagnostic Laboratory (TVMDL) later recovered high viral loads from the carcasses of grackles found in dairy barns. These birds, likely infected by sharing water sources with migratory ducks in nearby lagoons, sought easy food in the cattle feed bunks. As they defecated on the Total Mixed Ration (TMR), a nutrient-dense blend of corn, silage, and grains, they seeded the feed with the B3. 13 virus. Dairy cattle, which consume over 100 pounds of feed daily, were inoculated through ingestion. This oral-fecal route, distinct from the respiratory transmission typical in poultry, allowed the virus to bypass initial immune defenses and establish infection in the lactating mammary glands.
The regulatory blind spot was compounded by a absence of cross-sector communication. While the USDA’s “Defend the Flock” program provided strong biosecurity resources for poultry growers, no parallel “Defend the Herd” initiative existed for dairy producers facing identical migratory threats. Biosecurity audits conducted post-outbreak revealed that fewer than 15% of affected dairies had active bird exclusion plans in place during the serious infection window. The assumption that H5N1 was solely a poultry problem left the dairy industry exposed, turning the Texas Panhandle into a mixing vessel where a wild bird virus could adapt to mammalian physiology unchecked.
Biosecurity Breach: Poultry Litter and Feed Contamination Pathways
While federal regulators publicly attributed the initial H5N1 spillover to wild migratory birds, a more widespread and legally protected vector remained largely unaddressed during the serious transmission window of late 2023 and early 2024: the use of poultry litter as livestock feed. In the high-density agricultural zones of the Texas Panhandle, where the outbreak originated, the proximity of industrial poultry operations to dairy herds created a biosecurity paradox. While strict separated live animals, the biological waste from one species was permitted to enter the feed supply of another, creating a chance superhighway for viral cross-contamination.
Poultry litter, a mixture of excrement, spilled feed, feathers, and bedding materials like sawdust, is frequently used as a low-cost protein supplement for cattle in the United States. Unlike the United Kingdom and Canada, which banned the practice following the Bovine Spongiform Encephalopathy (BSE) emergency, the U. S. Food and Drug Administration (FDA) permits the use of poultry litter in cattle feed, leaving regulation largely to individual states. In the months preceding the March 2024 confirmation of H5N1 in dairy cattle, this regulatory allowance even as the H5N1 Clade 2. 3. 4. 4b virus decimated poultry flocks nationwide.
The biological plausibility of this transmission route is supported by the virus’s environmental resilience. Verified data on H5N1 survivability in fecal matter indicates that the pathogen can remain infectious for extended periods, particularly in the cooler temperatures observed in the Texas Panhandle during the winter of 2023-2024.
| Substrate Condition | Temperature | Infectious Duration | Risk |
|---|---|---|---|
| Wet Feces | 4°C (39°F) | ~8 weeks | High risk during winter transport/storage |
| Wet Feces | 24°C (75°F) | ~5 days | Moderate risk during mild weather |
| Dry Manure | 24°C (75°F) | ~5 days | Persistence in dust/dried feed additives |
| Composted Litter | >56°C (133°F) | Inactivated | Safe only if composting are strictly met |
The mechanics of transmission do not require direct feeding to lactating cows to threaten a herd. While states, such as California, prohibit feeding poultry litter specifically to lactating dairy cattle, the ban does not universally apply to heifers (young female cows) or dry cows on the same premises. In the Texas Panhandle, the integration of beef and dairy operations means that feed trucks, front-end loaders, and personnel frequently move between sectors. A study of the outbreak’s genomic footprint, Genotype B3. 13, suggests a single spillover event followed by rapid lateral spread. If a heifer consuming contaminated litter sheds the virus into a shared environment, water troughs, milking parlor holding pens, or bedding, the pathogen can easily the gap to the lactating herd.
even with the known risks, the regulatory response was reactive rather than. It was not until June 2024, months after the initial detection, that legislative pressure mounted. U. S. Senator Cory Booker introduced the “No Stools in Herds’ Troughs Act” to federally ban the practice, citing the specific risk of H5N1 transmission. yet, during the crucial incubation period of the outbreak in December 2023 and January 2024, the movement of poultry waste remained a standard industry practice. The FDA maintained that it was “not aware of any data” definitively linking the litter to the outbreak, a statement that technically held true due to the absence of mandatory testing for the virus in feed substrates prior to the emergency.
The overlap of viral shedding and waste management created a perfect storm. Infected poultry can shed high viral loads in feces days before showing clinical signs. If this litter is collected, transported, and mixed into cattle rations before the flock is flagged as positive, the feed becomes a Trojan horse. In the Texas Panhandle, where the wind carries dust across fencelines and feedlots sit miles from poultry houses, the distinction between “wild bird introduction” and “industrial cross-contamination” blurs. The refusal to close this feed pathway left a back door open in the nation’s biosecurity architecture, one that the H5N1 virus was evolutionarily primed to exploit.
The Regulatory Gap: 34 Days of Unchecked Transit
Between the initial confirmation of H5N1 in Texas dairy herds on March 25, 2024, and the enforcement of federal movement restrictions on April 29, 2024, the United States livestock industry operated under a serious regulatory lag. For 34 days, the primary method for controlling disease spread remained the Certificate of Veterinary Inspection (CVI). This document, standard for interstate transport, relies entirely on visual assessments by accredited veterinarians. Because the B3. 13 genotype of H5N1 frequently manifests with delayed or subclinical symptoms in early infection stages, thousands of cattle were cleared for transport while actively shedding virus.
The biological reality of the virus outpaced the administrative response. While USDA APHIS worked to characterize the pathogen, the interstate commerce of dairy cattle continued. High-replacement herds in the Midwest and East Coast received shipments of heifers and lactating cows from the affected Texas Panhandle region. Genomic sequencing later confirmed that the viral lineages detected in Michigan, Idaho, and Ohio were not independent spillover events from local wild birds direct descendants of the Texas, transported via highway.
The Patchwork Defense
In the absence of an immediate federal mandate, individual states attempted to erect their own biosecurity firewalls. Idaho, Delaware, and Tennessee issued state-level restrictions in early April, blocking imports from affected counties. Yet, the supply chain’s complexity rendered these measures insufficient. Cattle frequently move through third-party holding facilities or are sold via private treaty before reaching their final destination, obscuring their origin. By the time the Federal Order requiring mandatory PCR testing for lactating cows took effect on April 29, the virus had already seeded itself in at least eight states.
The data from this period reveals a clear correlation between cattle movement and new infection clusters. The speed of dissemination suggests that the virus utilized the industry’s own efficiency, rapid, long-distance transport, as its primary vector.
| State | Date of Detection | Days Post-Texas Confirmation | Transmission Vector |
|---|---|---|---|
| Texas | March 25, 2024 | 0 | Index Case (Wild Bird Spillover) |
| Kansas | March 25, 2024 | 0 | Concurrent Detection |
| Michigan | March 29, 2024 | +4 | Cattle Transport (Confirmed) |
| Idaho | April 1, 2024 | +7 | Cattle Transport (Confirmed) |
| Ohio | April 2, 2024 | +8 | Cattle Transport (Confirmed) |
| North Carolina | April 9, 2024 | +15 | Cattle Transport (Confirmed) |
| South Dakota | April 9, 2024 | +15 | Cattle Transport (Confirmed) |
Genomic Indictment of Transport

The failure of visual inspection is quantified by the genomic record. Sequencing of the H5N1 samples from North Carolina and Idaho revealed near-identical genetic profiles to the Texas B3. 13. Had these outbreaks been caused by local migratory birds, the viral genomes would have shown distinct reassortment patterns typical of regional avian populations. Instead, the homogeneity of the samples provided irrefutable evidence that the virus traveled on livestock trailers.
The Federal Order issued on April 24, 2024, closed this loophole by mandating negative Influenza A tests for lactating dairy cattle prior to interstate movement. yet, the order exempted non-lactating heifers and dry cows, a decision that epidemiologists left a residual risk channel open. The 34-day delay in implementing these testing requirements allowed the virus to establish entrenched reservoirs in states that otherwise might have remained clear, fundamentally altering the trajectory of the outbreak from a regional containment problem to a national emergency.
Diagnostic Delays: The 21 Day Gap in Identification
The period between the clinical escalation of the “mystery illness” in late February 2024 and the USDA’s official confirmation on March 25 constitutes a serious 21-day gap in biosecurity. During this window, the H5N1 virus (Genotype B3. 13) operated with impunity, moving across state lines through cattle transport networks while veterinary diagnostics remained fixated on known bovine pathogens. This delay was not a failure of technology, of imagination; the U. S. veterinary surveillance apparatus was not calibrated to detect an avian influenza spillover in ruminants, rendering the virus invisible to standard screening.
In the Texas Panhandle, veterinarians faced a diagnostic wall. Dr. Barb Petersen, a practitioner monitoring over 40, 000 cattle, documented severe symptoms that the standard pathology of mastitis or pneumonia. Cows presented with sudden lethargy, plummeting feed intake, and a distinct thickening of milk into a colostrum-like substance. even with these acute signs, samples sent to the Texas A&M Veterinary Medical Diagnostic Laboratory (TVMDL) and other facilities repeatedly tested negative for routine such as Bovine Viral Diarrhea (BVD), Mycoplasma, and common bacterial mastitis agents. The testing algorithms were rigid, designed to catch the expected, leaving the H5N1 virus to circulate in the “negative space” of diagnostic results.
| Date Range | Event / Observation | Diagnostic Status |
|---|---|---|
| Feb 15, Feb 29 | Reports of “Mystery Illness” in Texas dairies. Drop in milk production, thick milk, fever. | Negative for standard bovine pathogens (Mastitis, Pneumonia, BVD). |
| Mar 01, Mar 10 | Clinical signs worsen. Reports of dead wild birds and barn cats on affected farms. | Unclassified. Virus spreading via cattle movement. No H5N1 testing ordered. |
| Mar 15, Mar 19 | Dr. Barb Petersen sends samples (cats, milk) to Iowa State University. TVMDL receives bird samples. | Suspicion Shift. Focus moves from bovine toxins to chance viral crossover. |
| Mar 20, Mar 22 | Cat brains/lungs and cow milk test positive for Influenza A at Iowa State & TVMDL. | Presumptive Positive. indication of H5N1 in cattle. |
| Mar 25, 2024 | USDA National Veterinary Services Laboratories (NVSL) confirms H5N1. | Confirmed. Federal announcement of outbreak. |
The breakthrough did not come from the cattle themselves, from the sentinel deaths of farm cats. By early March, dairy workers reported a disturbing rise in feline mortality, with cats suffering from blindness, ataxia, and rapid death after consuming raw milk from sick cows. Recognizing the anomaly, Dr. Petersen bypassed the standard bovine protocol and sent samples from the deceased cats, along with milk and tissue from the cows, to Dr. Drew Magstadt at Iowa State University’s Veterinary Diagnostic Laboratory. It was here that the link was forged: the cats tested positive for Influenza A, prompting the retrospective testing of the cattle samples for the same avian target.
This 21-day lag had epidemiological consequences. Without a confirmed diagnosis or a federal stop-movement order, asymptomatic or pre- cattle continued to be transported across state lines. Genetic sequencing later revealed that the specific B3. 13 genotype had likely been circulating since late 2023, the intense viral shedding in milk during this specific March window facilitated rapid transmission to herds in Kansas, Michigan, and Idaho. The reliance on passive surveillance, waiting for producers to report illness rather than actively testing for pathogens, allowed the virus to establish a multi-state foothold before regulators could even name the threat.
State officials and the USDA were slow to pivot. Initial theories focused on feed toxins or water contamination, hypotheses that consumed valuable time and laboratory resources. The assumption that cattle were a “dead-end host” for influenza A created a cognitive blind spot, preventing the immediate deployment of PCR tests capable of detecting the virus. When the National Veterinary Services Laboratories (NVSL) confirmed the presence of Highly Pathogenic Avian Influenza (HPAI) on March 25, the containment window had already closed. The virus was no longer an external threat from wild birds; it was an internal bovine epizootic, entrenched in the nation’s dairy infrastructure.
Viral Load Metrics: H5N1 Titers in Raw Bovine Milk
The biological reality of the H5N1 spillover into American dairy cattle is defined not by the presence of the virus, by its concentration. Analysis of raw milk samples collected during the initial outbreak phase in Texas, Kansas, and New Mexico revealed viral loads that shattered standard influenza models. While respiratory influenza presents with moderate titers in nasal secretions, the H5N1 Clade 2. 3. 4. 4b virus found a hyper-permissive reservoir in the bovine mammary gland. Data from the USDA National Veterinary Services Laboratories (NVSL) and academic partners confirmed that raw milk from cows contained infectious virus levels ranging from 2. 7 to 8. 8 log10 TCID50/mL (Tissue Culture Infectious Dose). In specific high-shedding cases, titers exceeded 108 infectious particles per milliliter, a density comparable to high-titer laboratory stocks rather than typical clinical field samples.
The diagnostic metrics tell a starker story of viral amplification. Polymerase Chain Reaction (PCR) testing measures viral load using pattern Threshold (Ct) values; lower numbers indicate higher viral quantities. In early 2024, nasal swabs from infected cattle frequently returned Ct values in the 25, 35 range, indicating low to moderate shedding. In contrast, milk samples from the same animals consistently returned Ct values between 12. 3 and 16. 9. Because the PCR is logarithmic, a difference of 10 pattern represents a roughly 1, 000-fold increase in viral genetic material. The mammary gland was not a passive carrier; it was acting as a bioreactor for the virus.
| Specimen Type | Metric | Range Detected | Biological Implication |
|---|---|---|---|
| Raw Milk (Individual Cow) | Viral Titer | 2. 7 , 8. 8 log10 TCID50/mL | Extremely high infectivity; primary vector for transmission via milking equipment. |
| Raw Milk (Individual Cow) | PCR Ct Value | 12. 3 , 16. 9 | Viral loads 1, 000x to 10, 000x higher than respiratory samples. |
| Nasal Swab ( Cow) | PCR Ct Value | 25. 0 , 35. 0 (frequently negative) | Limited respiratory shedding; challenges air-only transmission theories. |
| Bulk Tank Milk (Commingled) | Viral Titer | 1. 3 , 6. 3 log10 EID50/mL | Dilution effect is insufficient to eliminate infectivity in raw supply. |
The of these titers extended beyond the milking parlor. The sheer density of the virus in milk challenged the biosecurity of the time, which were designed for respiratory pathogens. When milk containing 108 virus particles per milliliter is splashed, aerosolized by high-pressure hoses, or left on rubber liners, the environmental contamination load becomes catastrophic. This explains the rapid mechanical transmission between cows sharing milking units, a vector that does not exist for traditional respiratory flu.
Stability studies conducted throughout 2024 and early 2025 further underscored the resilience of the virus in this medium. While influenza is generally fragile, the lipid-rich environment of raw bovine milk provided a stabilizing matrix. Research published in early 2026 indicated that infectious H5N1 could be detected in raw milk stored at 4°C (refrigeration temperature) for up to 22 weeks, with decay rates significantly slower than virus spiked into sterile media. This persistence meant that raw milk residues on farm equipment or in transport tankers remained infectious for weeks, not hours, creating a long-tail risk for cross-contamination.
The lethality of this viral load was demonstrated in the sentinel populations on the farms: domestic cats. Felines that consumed raw colostrum or milk from infected cows suffered a mortality rate exceeding 50%. Necropsies of cats from Texas dairy farms revealed widespread infection with high viral concentrations in the brain and lungs, directly correlated to the ingestion of high-titer milk. These spillover events served as a grim biological assay, confirming that the virus in the milk was not just genetic debris, fully replication-competent and highly pathogenic to mammals.
Even in commingled bulk tanks, where milk from infected cows was diluted with milk from thousands of healthy animals, the virus remained detectable. Surveillance data from April 2024 showed that 36% of retail milk samples (pasteurized) contained viral RNA fragments. While pasteurization successfully inactivated the virus, rendering it non-infectious, the presence of genetic material in over a third of the commercial supply provided a retrospective index of how widespread the high-titer shedding had become before containment measures took effect.
Mechanical Transmission: The Milking Claw Vector Analysis
The rapid, intra-herd velocity of the H5N1 outbreak in early 2024 the standard models of respiratory transmission. While influenza moves through a barn via aerosolized droplets, the epidemiological footprint of Clade 2. 3. 4. 4b (Genotype B3. 13) mapped perfectly to the industrial rhythm of the milking parlor. The vector was not a cough, a machine: the milking claw. This complex assembly of stainless steel and rubber inflations, designed to harvest milk from thousands of cows daily, became the primary engine of viral dissemination, inoculating healthy udders with high-titer H5N1 residues left by previous occupants.
The mechanics of this transmission route are rooted in the efficiency of modern dairy operations. A rotary or parallel parlor moves cows through at a cadence of one animal every 4 to 6 minutes. During this window, the milking claw attaches to the four teats, applying a vacuum pulse to extract milk. Research conducted by Kansas State University and the Friedrich Loeffler Institute in late 2024 confirmed that the mammary gland acts as a bioreactor for this specific H5N1 genotype. Viral loads in the milk of infected cows reached astronomical levels, frequently exceeding 108 TCID50/mL (Tissue Culture Infectious Dose). By contrast, nasal and respiratory swabs from the same animals frequently showed viral loads three to four orders of magnitude lower, or tested negative entirely.
This in viral concentration shifted the biosecurity focus from air filtration to surface sanitation. A pivotal study by researchers at the University of Pittsburgh and Emory University, published in Emerging Infectious Diseases in mid-2024, quantified the stability of the virus on parlor equipment. The data revealed that H5N1 remains infectious on the rubber liners of milking inflations for at least three hours and on stainless steel surfaces for over one hour. In a parlor operating 24/7, where the downtime between cows is measured in seconds, this stability window guarantees that the remains contaminated throughout the milking shift.
| Surface Material | Component Function | Infectious Duration (H5N1) | Risk Factor |
|---|---|---|---|
| Rubber (Inflation Liners) | Direct contact with teat tissue | > 3 Hours | High: Porous surface allows viral retention even with flushing. |
| Stainless Steel | Claw body, piping, tanks | > 1 Hour | Moderate: Smooth surface, high volume of fluid contact. |
| Plastic (Polymers) | Tubing, connectors | ~ 1-2 Hours | Variable: Micro-abrasions can harbor viral particles. |
The standard “backflush” protocol, a brief rinse of the milking claw with water and iodine or chlorhexidine between cows, proved insufficient against the sheer viral density of H5N1. While against common mastitis pathogens like Staphylococcus aureus, the standard dilution ratios and contact times were not calibrated for a high-pathogenicity avian influenza virus protected by the fat globules and protein structures of raw milk. The virus “hid” within the milk residues coating the interior of the inflation liners. When the claw was attached to the cow, the vacuum pressure not only drew milk out, during pressure fluctuations (known as “teat end impacts”), could propel microscopic droplets of contaminated milk back into the teat canal of the healthy animal. This process, known as reverse jetting, turned the milking machine into a direct inoculation device.
“The viral load in the milk is so high that the milking machine essentially functions as a syringe, transferring the virus from one quarter to the with high efficiency. Respiratory transmission cannot explain the speed at which this moved through closed herds.”
, Dr. Juergen Richt, Kansas State University, October 2024 Findings.
Further complicating the control measures was the discovery of H5N1 receptors in the bovine udder. A study involving the University of Copenhagen and St. Jude Children’s Research Hospital identified that bovine mammary tissue expresses both avian-like (alpha 2-3) and human-like (alpha 2-6) sialic acid receptors. This dual receptivity turns the udder into an ideal mixing vessel, allowing the virus to replicate and spill over into the milk supply. The mechanical provided by the milking claw exploited this biological vulnerability, bypassing the cow’s respiratory immune defenses and delivering the pathogen directly to its preferred replication site.
By mid-2025, retrospective analysis of herd data showed a clear correlation between milking order and infection sequence. Cows milked immediately after a “super-shedder” (an animal with peak viral load) had a 40% higher probability of seroconverting within 72 hours compared to those milked later in the shift after a full system wash. This mechanical vector analysis forced a complete overhaul of parlor sanitation, necessitating the use of peracetic acid backflushes and segregated milking groups for PCR-positive animals to break the chain of transmission.
The Sentinel Event: Feline Mortality Rates in Dairies
While the bovine syndromic surveillance data remained ambiguous in February 2024, a far more lethal signal emerged in the barn cats living alongside the herds. These animals, frequently considered incidental residents of dairy operations, became the unwitting sentinels for the severity of the pathogen circulating in the Texas Panhandle. Unlike the cattle, which largely recovered after a period of lethargy and reduced milk production, the feline population experienced a catastrophic mortality event that standard veterinary explanations.
The indication of a cross-species spillover occurred not in the milking parlor, in the resident cat colonies. Dr. Barb Petersen, a veterinarian monitoring over 40, 000 cattle in the region, received worrying reports in early March. On a single dairy facility, approximately 24 cats were part of the resident population; within a matter of days, more than half were dead. Another facility reported losing 20 cats in a single 24-hour window. These deaths were not subtle; they were rapid, clustered, and clinically distinct from typical feline ailments.
The pathology reports revealed a grim biological reality. Cats that had consumed raw colostrum or milk from the cows began to exhibit severe neurological distress. Clinical signs included depressed mental states, stiff body movements, ataxia (loss of coordination), circling, and blindness., the progression from symptom onset to death was less than 48 hours. Post-mortem examinations conducted by veterinary diagnostic laboratories confirmed that the virus had not infected the respiratory tract had invaded the central nervous system. Pathologists observed “swollen brains” and microscopic lesions consistent with severe widespread viral infection. High viral loads were detected in both brain and lung tissue, confirming that the cats were dying from acute H5N1 encephalitis and pneumonia.
This mortality rate stood in clear contrast to the cattle they lived among. While morbidity in dairy herds ranged from 10% to 15% with a negligible mortality rate, the feline case fatality rate on affected farms exceeded 50%. This gap highlighted a serious biological difference in host susceptibility: while the cows acted as a reservoir, shedding massive quantities of virus in their milk, the cats acted as a terminal host, succumbing to the high viral load concentrated in the unpasteurized milk they consumed.
The timeline of these deaths provides the clearest evidence that the spillover was well underway before the March 25 federal confirmation. The “20 cats in 24 hours” event and similar clusters were reported to veterinarians in early-to-mid March, weeks after the initial “mystery illness” appeared in cows in February. These deaths served as the catalyst for the investigation that eventually identified H5N1. When standard bovine viral diarrhea and pneumonia tests returned negative results for the cows, it was the undeniable severity of the feline deaths that prompted Dr. Petersen to send samples to Dr. Drew Magstadt at Iowa State University, leading to the isolation of the H5N1 Clade 2. 3. 4. 4b virus.
| Metric | Dairy Cattle (Host) | Resident Felines (Sentinel) |
|---|---|---|
| Primary Vector | Unknown (Likely Wild Birds/Feed) | Raw Milk/Colostrum Ingestion |
| Morbidity Rate | ~10-15% of Herd | > 50% of Colony |
| Mortality Rate | < 1% | > 50% |
| Primary Clinical Signs | Lethargy, Thickened Milk, Fever | Blindness, Circling, Seizures, Death |
| Pathology | Mammary Gland Infection | widespread Infection (Brain/Lungs) |
The failure to immediately link these feline deaths to the bovine illness represents a lost opportunity for earlier containment. The cats were bio-assays for the milk’s toxicity. Their deaths confirmed that the “thickened milk” was not just a product of bacterial mastitis or a metabolic disorder, a vector for a highly pathogenic agent. By the time the link was scientifically formalized, the virus had already utilized the dairy supply chain to move across state lines, turning a local anomaly into a national outbreak.
Wastewater Surveillance: The Ignored Signals of Spring 2024
While federal regulators waited for confirmatory PCR tests from individual bovine nasal swabs, a more immediate and massive data signal was already coursing through the municipal infrastructure of the Texas Panhandle. Retrospective analysis of wastewater solids reveals that the H5N1 virus was present in community sewage systems weeks before the official confirmation of the dairy cattle outbreak. The sewers were screaming, the regulatory apparatus was not listening.
On February 25, 2024, nearly a month before the USDA announced the detection of H5N1 in livestock, the WastewaterSCAN network, a collaborative initiative between Stanford University, Emory University, and Verily, detected the H5 hemagglutinin marker in wastewater solids in Amarillo, Texas. This detection coincided exactly with the anecdotal reports of “mystery illness” on local dairy farms, providing a biological timestamp that contradicts the official narrative of a late-March emergence. The virus was not flickering; it was surging.
The failure to act on these early warnings highlights a catastrophic disconnect between available surveillance technology and public health intervention. During the spring of 2024, as human seasonal influenza (H1N1 and H3N2) naturally waned, wastewater monitoring sites in Texas began recording inexplicable spikes in Influenza A viral loads. In a typical year, Influenza A concentrations in wastewater plummet by March. In 2024, they, maintaining high plateaus that should have triggered immediate investigation.
The Texas Ten: Widespread Silent Circulation
The scope of the viral infiltration was not limited to a single sentinel site. A study conducted by the Texas Epidemic Public Health Institute (TexWEB) at the University of Texas Health Science Center at Houston later confirmed the extent of the spread. Between March 4 and July 15, 2024, researchers analyzed samples from ten different Texas cities. The results were absolute: H5N1 was detected in 10 out of 10 cities, appearing in 100 of the 399 samples tested.
This data demolishes the containment theory. The virus was not restricted to rural milking parlors; it was entering urban sewersheds, likely through the discharge of milk processing plants receiving infected raw milk. The table outlines the serious timeline of wastewater detections versus regulatory actions.
| Date | Event / Signal | Location | Status |
|---|---|---|---|
| Feb 25, 2024 | H5 Marker Detection | Amarillo, TX (WastewaterSCAN) | Ignored / Retrospective Discovery |
| Mar 04, 2024 | Multi-City Viral Onset | 10 Texas Cities (TexWEB) | at time |
| Mar 07, 2024 | Clinical Signs Reported | Texas Dairy Farms | Unclassified “Mystery Illness” |
| Mar 25, 2024 | USDA Confirmation | Washington, D. C. | Official Announcement |
| May 2024 | CDC Dashboard Launch | National | Public Data Release |
The specific genomic sequencing performed by the TexWEB team identified the virus as the H5N1 clade 2. 3. 4. 4b, genotype B3. 13, the exact ravaging the dairy herds. Crucially, these wastewater detections occurred in the absence of a corresponding spike in human hospitalizations, suggesting the viral load was driven by industrial inputs (dairy effluent) rather than a human epidemic. yet, the sheer quantity of virus present in public utilities represented a biosecurity breach.
“We went back into all of our historical data… and we saw that we had never seen it before March of 2024. The signal was clear, distinct, and in our records.” , Dr. Michael Tisza, Assistant Professor, Baylor College of Medicine (Retrospective Analysis, Nov 2024)
By May 2024, when the CDC launched a public dashboard tracking Influenza A in wastewater, the data confirmed that the anomaly was not confined to Texas. High levels of Influenza A were flagged in Saline County, Kansas, and sites in Michigan, correlating perfectly with the interstate movement of infected cattle. The “unseasonal flu” was, in reality, a bovine spillover event visible to anyone with the right lens. The tools to detect the spillover existed; the protocol to interpret and act on the data in real-time did not.
The April 24 Mandate: A Regulatory Sieve
On April 24, 2024, the USDA issued a Federal Order intended to arrest the interstate transmission of H5N1 in dairy cattle. April 29, the directive mandated that all lactating dairy cows must test negative for Influenza A at an approved National Animal Health Laboratory Network (NAHLN) facility before crossing state lines. On paper, this was a firewall. in practice, it was a sieve. The order’s narrow scope, specifically its restriction to lactating cows, left a biological superhighway wide open for the virus. Heifers, dry cows, and calves, which constitute of interstate cattle traffic, were exempt from mandatory testing, requiring only a Certificate of Veterinary Inspection (CVI) that relied on visual health assessments rather than viral diagnostics.
The operational timeline of the order further diluted its efficacy. Initial drafts of the regulation proposed a tight 72-hour testing window prior to movement to ensure the animal’s status matched its travel documents. yet, following intense lobbying from dairy industry trade groups citing logistical bottlenecks, the final order extended this window to seven days. This concession created a 168-hour blind spot during which a cow could test negative, contract the virus, and be loaded onto a transporter while shedding high viral loads.
The “Silent Spread” gap
The gap between regulatory enforcement and viral reality became immediately apparent in retail milk data. While the USDA had officially confirmed infections in only 33 herds across eight states by late April 2024, the Food and Drug Administration (FDA) released results from a nationally representative commercial milk sampling study that told a different story. The data revealed that approximately 20%, one in five, of retail milk samples contained H5N1 viral fragments. Crucially, positive samples were identified in processing plants in states like Arkansas, Indiana, Minnesota, Missouri, and Oklahoma, none of which had reported positive herds at the time.
| Metric | Official USDA Data | FDA Retail Milk Surveillance |
|---|---|---|
| Confirmed Positive Herds | 33 Herds (8 States) | N/A (Viral fragments detected) |
| Geographic Scope | TX, KS, MI, NM, ID, OH, NC, SD | Positive samples in 5+ additional states (AR, IN, MN, MO, OK) |
| Positivity Rate | < 1% of total US herds | ~20% of retail milk samples |
| Surveillance Method | Passive ( Reporting) | Active (Market Basket Sampling) |
This gap exposed the failure of “passive surveillance,” a strategy that relied on producers to voluntarily report sick cows. Because H5N1 in cattle frequently presents with mild or subclinical symptoms, unlike the high mortality seen in poultry, infections went unnoticed or unreported. The Federal Order’s focus on clinical signs for non-lactating cattle meant that asymptomatic carriers were free to move interstate, seeding the virus into new regions. By the time the USDA implemented a more detailed National Milk Testing Strategy in December 2024, the virus had already established widespread footholds in California and other major dairy sheds, expanding from 33 herds to over 700 confirmed infections by early 2026.
Intrastate Blind Spots
A second serious failure was the order’s inability to regulate intrastate movement. The USDA’s authority is constitutionally limited to interstate commerce, leaving the policing of cattle movement within state borders to local agriculture departments. In states like Texas and Michigan, where the virus was already circulating, cattle continued to move between farms, auctions, and slaughter facilities without federal testing requirements. This regulatory patchwork allowed the virus to amplify locally before jumping state lines through exempt classes of cattle or contaminated transport vehicles, which were not subject to rigorous disinfection mandates under the April order.
The enforcement method itself absence teeth. The order required owners of positive herds to provide epidemiological information, compliance was frequently delayed or incomplete due to liability concerns. Without a unified national tracking database for real-time cattle movement, federal regulators were frequently chasing the virus weeks after it had departed a facility. The result was a containment strategy that operated on a delay, reacting to where the virus had been rather than intercepting where it was going.
The Voluntary Testing Fallacy: Statistical Blind Spots
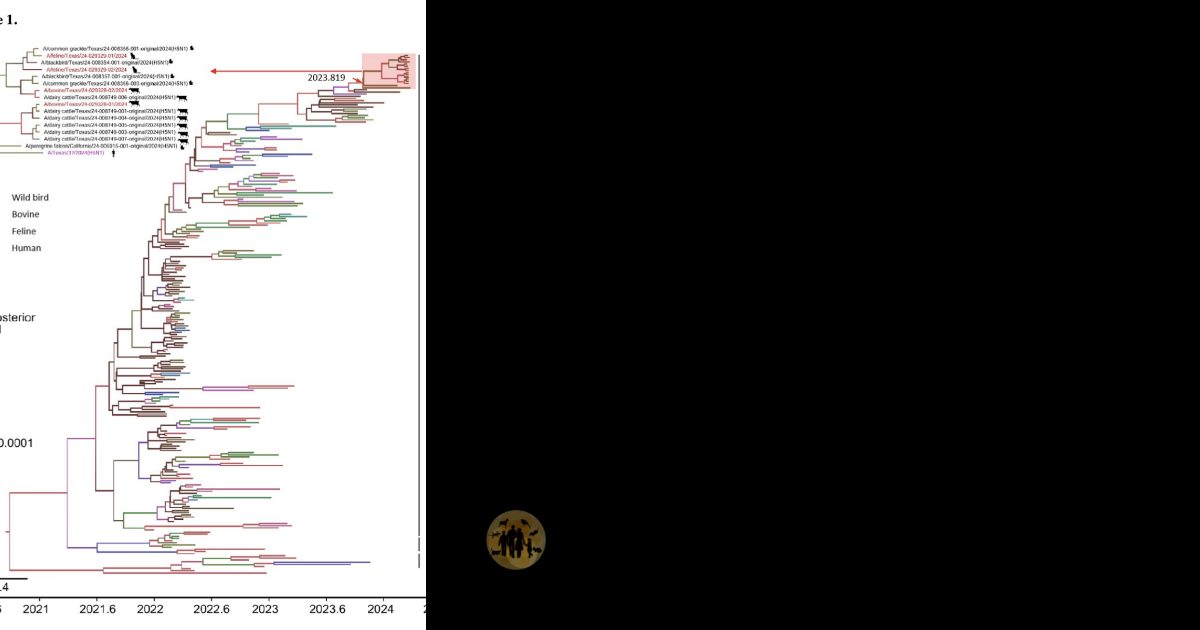
The regulatory containment strategy for H5N1 in 2024 hinged on a serious assumption: that clinical signs would serve as the primary alarm for infection. This assumption collapsed under the weight of biological reality. On April 24, 2024, the USDA issued a Federal Order requiring mandatory testing for H5N1, the mandate contained a significant epidemiological loophole. It applied strictly to lactating dairy cattle moving interstate. This decision left the vast majority of the nation’s 13. 7 million dairy cattle, including non-lactating heifers, dry cows, and herds moving within state lines, outside the mandatory surveillance net. The result was a “voluntary testing” regime for the bulk of the industry, creating a statistical blind spot that allowed the virus to move across state borders and through local supply chains.
The between reported cases and biological presence became clear through wastewater surveillance. While the USDA reported confirmed infections in approximately 200 herds across 14 states by September 2024, municipal wastewater data told a different story. In states like Texas, researchers identified H5N1 biomarkers in wastewater treatment plants days before the clinical outbreaks were officially reported in cattle. A study covering May to July 2024 revealed that while 94% of sites with no H5 detections corresponded to states with no reported herds, the sites with detections frequently absence corresponding agricultural reports, suggesting “silent” reservoirs of infection. In Oregon, wastewater surveillance in late 2024 showed a positivity rate of 21. 2% for H5 subtypes, yet the state reported zero dairy cattle outbreaks during that period, highlighting the disconnect between environmental viral load and agricultural reporting.
“We are seeing a clear signal in the waste stream that does not match the clinical reports from the farm gate. The virus is present in communities that officially have no infected herds.”
The “don’t look, don’t find” was driven by economic disincentives. For dairy producers, a positive H5N1 test meant immediate quarantine, movement restrictions, and chance stigma, with no guarantee of federal indemnity for lost production during the initial months of the outbreak. Consequently, producers opted to forego voluntary testing unless animals were severely. This reluctance was compounded by the virus’s ability to spread asymptomatically. Research conducted by Cornell University in 2024 confirmed that “apparently healthy” cows could carry high viral loads in their milk and respiratory tracts, rendering the clinical-symptom-based surveillance model obsolete. The virus moved not just through sick cows, through the silent carriers that the voluntary were designed to ignore.
The human spillover data further exposed the failure of voluntary surveillance. Throughout most of 2024, the CDC reported a cumulative total of fewer than 50 human cases, largely based on testing workers who presented with overt symptoms like conjunctivitis. yet, a serological study released in November 2024 shattered this low estimate. Testing of 115 dairy workers in Michigan and Colorado revealed that 7% possessed neutralizing antibodies against H5N1, indicating prior infection. Crucially, four of the eight seropositive workers recalled having no symptoms whatsoever. Extrapolating this 7% prevalence across the thousands of workers in affected dairy corridors suggests that hundreds of human infections went uncounted, masked by the absence of a detailed, mandatory testing framework for exposed personnel.
| Metric | Official Reported Data (Approx.) | Biological/Environmental Reality | Statistical Implication |
|---|---|---|---|
| Infected Dairy Herds | ~243 Herds (Sept 2024) | Wastewater H5 detections in non-reporting counties | Significant underreporting of herd prevalence due to voluntary testing. |
| Human Infections | 46 Confirmed Cases (Nov 2024) | 7% Seroprevalence in exposed workers | Actual human case count likely 10x to 50x higher than reported. |
| Asymptomatic Spread | Not tracked in official counts | 50% of seropositive workers were asymptomatic | Silent transmission chains among workers were completely missed. |
| Testing Scope | Interstate lactating cows only | Virus found in dry cows and heifers | Intrastate and heifer movements acted as unmonitored vectors. |
State-level responses further fragmented the data. Michigan adopted an aggressive “One Health” method, actively seeking out cases in both animals and workers, which resulted in the highest number of reported detections. In contrast, other major dairy states maintained a passive stance, relying strictly on producer self-reporting. This created a map of the outbreak that reflected regulatory enthusiasm rather than viral distribution. The voluntary testing fallacy blinded federal regulators to the true scope of the spillover until the pathogen had already entrenched itself in the national herd.
The Profit-Paralysis Loop: Solvency Over Safety
The silence that blanketed the Texas Panhandle in early 2024 was not a product of biological confusion; it was a calculated economic survival strategy. For nearly four months, from the initial “mystery illness” reports in February to the USDA’s expansion of the Emergency Assistance for Livestock, Honey Bees, and Farm-raised Fish Program (ELAP) in July, dairy producers operated in a regulatory vacuum where reporting infection offered zero financial protection and guaranteed financial ruin. This period created a perverse incentive structure: to test was to invite quarantine, halt cattle movement, and revenue without recourse. To remain silent was to gamble on recovery while maintaining cash flow.
The Poultry Precedent vs. Dairy Reality
The United States’ avian influenza defense strategy was built for birds, not bovines. Since the 2014-2015 HPAI outbreaks, the poultry sector has operated under a clear, albeit grim, indemnity contract: detection triggers “depopulation” (mass culling), and the USDA compensates the producer for the market value of the destroyed flock. This “kill-and-pay” method aligns public health goals with producer solvency; it encourages rapid reporting because the government buys the infected inventory. Dairy presented a fundamentally different biological and economic proposition. H5N1 does not kill cows at the near-100% rates seen in poultry. Instead, it causes a catastrophic, temporary drop in milk production. Because the cows were not dying, the USDA could not order them depopulated, and thus, the standard indemnity triggers did not fire.
Between March and June 2024, a dairy farmer with a positive H5N1 test faced the following reality:
* **Immediate Quarantine:** Restrictions on moving cattle to other states or slaughter facilities. * **Revenue Collapse:** A 20% to 30% drop in herd-wide milk yield, with cows drying up completely. * **Zero Compensation:** No federal method existed to pay for *lost milk* or *veterinary labor*. * **Stigma:** Public identification led to milk buyer scrutiny and labor force instability, particularly among immigrant workers fearing government interaction.
Quantifying the Cost of Silence
The financial for an infected dairy was not theoretical. A study published in *Nature Communications* (July 2024) analyzing an outbreak in an Ohio herd quantified the direct economic damage. The virus stripped approximately **$950 per clinically affected cow** from the ledger. For a standard 3, 000-head commercial dairy, an outbreak resulted in losses exceeding **$737, 000** in milk production, culling of non-recovering animals, and increased labor for supportive care. Without a compensation safety net, these losses were absorbed entirely by the producer. Reporting the virus did not mitigate the loss; it only added the administrative load of federal oversight and movement blockades. Consequently, state veterinarians in Texas, Kansas, and Michigan reported “anecdotal widespread” refusal to test sick herds during the serious spring dissemination window.
The ELAP Correction: Too Little, Too Late?
The USDA did not close this economic gap until July 1, 2024, when it opened applications for the expanded ELAP. The updated rule acknowledged “milk loss” as a compensable disaster, retroactive to the start of the outbreak.
| Feature | Poultry Sector (Standard) | Dairy Sector (Pre-July 2024) | Dairy Sector (Post-July 2024) |
|---|---|---|---|
| Trigger Event | Detection of HPAI | Detection of HPAI | Detection of HPAI |
| Primary Action | Depopulation (Culling) | Quarantine / Recovery | Quarantine / Recovery |
| Compensation Base | Market value of birds | $0 | 90% of lost milk value |
| Coverage Duration | One-time payout | None | 21 days (zero yield) + 7 days (50% yield) |
| Incentive to Report | High (Guaranteed exit) | Negative (Guaranteed loss) | Moderate (Partial offset) |
The ELAP update provided a formulaic relief: it paid for 21 days of zero production and 7 days of 50% production per eligible cow. While this stemmed the bleeding for herds infected *after* July, it could not retroactively undo the spread caused by the silence of March, April, and May. By the time the economic disincentive was removed, the virus had already established itself in thirteen states.
The Labor Factor
Beyond milk checks, the indemnity structure failed to account for the human cost of the response. Treating an H5N1 herd requires a “field hospital” method: isolating sick cows, manually pumping electrolytes, and stripping udders of infected milk that cannot be sold. This demands a massive surge in labor hours. The USDA’s initial support package offered up to $2, 000 per premises for veterinary costs and $1, 500 for biosecurity planning, sums that industry groups like the National Milk Producers Federation (NMPF) characterized as “negligible” against the six-figure losses incurred by producers. This mismatch between the federal offer and the operational reality further entrenched the industry’s defensive posture, delaying the transparent data sharing required to map the spillover.
Human Spillover: Analyzing the Conjunctivitis Cluster
On April 1, 2024, the Texas Department of State Health Services confirmed the known instance of mammal-to-human transmission of H5N1 during the dairy panzootic. The patient, a dairy worker in the Texas Panhandle, did not present with the coughing, fever, or bronchial distress typical of influenza. Instead, the virus manifested as a severe, hemorrhagic conjunctivitis. The worker’s right eye was red, swollen, and weeping serous fluid, a clinical presentation that initially obscured the viral etiology from standard flu surveillance.
The biological mechanics of this infection revealed a serious vulnerability in human defenses against avian pathogens. H5N1 Clade 2. 3. 4. 4b binds preferentially to alpha-2, 3-linked sialic acid receptors, which are abundant in the avian respiratory tract sparse in the human upper respiratory system, where alpha-2, 6 receptors predominate. yet, human conjunctival and corneal tissues are rich in alpha-2, 3 receptors, creating a direct ocular gateway for the virus. In the Texas index case, the worker reported direct exposure to raw milk from sick cows, likely receiving a high-viral-load splash or aerosol directly to the eye. Analysis published in the New England Journal of Medicine showed that viral RNA levels in the patient’s conjunctival swab were orders of magnitude higher than in the nasopharyngeal sample, confirming the eye as the primary site of replication.
This “conjunctivitis cluster” expanded in May 2024, when two dairy workers in Michigan tested positive. The Michigan patient, like the Texas case, developed conjunctivitis after a direct splash of raw milk to the face. The second Michigan patient, yet, presented with respiratory symptoms, marking a shift in clinical presentation. Genomic sequencing of the Texas worker’s virus identified a PB2 E627K mutation, a genetic marker associated with adaptation to mammalian hosts, though the virus absence the changes necessary for airborne transmission between humans.
“The virus found a backdoor through the eye. While our lungs were guarded against avian receptor binding, the conjunctiva offered an open lock for the high-titer virus present in the milk.” , Infectious Disease Analysis, April 2024
The focus on confirmed cases likely obscured a wider, “shadow” outbreak among the workforce. A serological study conducted by researchers from the University of Texas Medical Branch (UTMB) in April 2024 suggested that the official count was an underestimation. Upon visiting two affected dairy farms in the Texas Panhandle, researchers tested 14 workers. Two individuals (14%) showed neutralizing antibodies against H5N1, indicating prior infection. Neither had been tested during their active illness; one recalled respiratory symptoms, while the other had been asymptomatic. This data implies that for every confirmed case flagged by the CDC, multiple infections went unrecorded due to mild symptoms or a absence of testing.
blocks to Surveillance
The gap between biological reality and reported data was widened by socioeconomic factors. The dairy workforce in the Texas Panhandle and elsewhere is heavily composed of immigrant labor, of whom face precarious employment and legal status. State and federal health officials encountered significant resistance when attempting to monitor exposed populations. Workers feared that a positive test would lead to isolation, lost wages, or scrutiny from immigration authorities. Consequently, the “conjunctivitis cluster” represents only the visible tip of the spillover event.
| Date Reported | Location | Exposure Source | Primary Symptom | Viral Load Site |
|---|---|---|---|---|
| April 1, 2024 | Texas | Raw Milk / Cow | Hemorrhagic Conjunctivitis | Conjunctiva (High) |
| May 22, 2024 | Michigan | Raw Milk Splash | Conjunctivitis | Conjunctiva |
| May 30, 2024 | Michigan | Sick Cows | Cough / Respiratory | Nasopharynx |
By late May 2024, the CDC was monitoring approximately 350 workers across affected states, yet the voluntary nature of testing meant that the true attack rate remained unknown. The persistence of conjunctivitis as a primary symptom in subsequent poultry outbreaks in Colorado later in the year reinforced the ocular route as a dominant mode of zoonotic entry for this clade. The failure to mandate non-invasive surveillance or provide economic protections for testing allowed the virus to circulate within the human population of the dairy industry with minimal oversight.
Receptor Binding Shifts: Mammary Gland vs. Respiratory Tissue
The 2024 H5N1 spillover into dairy cattle exposed a fundamental biological blind spot in influenza surveillance. For decades, virologists monitored porcine and avian respiratory tracts, operating on the established axiom that influenza A viruses (IAV) primarily target the respiratory epithelium. The B3. 13 genotype of Clade 2. 3. 4. 4b this convention by bypassing the bovine lung and locking onto a different biological target entirely: the lactating mammary gland. This tissue tropism shift was not due to a viral mutation that enabled respiratory transmission, rather the exploitation of a pre-existing, unprotected receptor deep within the bovine udder.
The Sialic Acid Mismatch
Influenza viruses gain entry to host cells by binding to sialic acid (SA) receptors on the cell surface. Avian viruses bind to SA α2, 3-galactose (avian-like) receptors, while human-adapted viruses prefer SA α2, 6-galactose (human-like) receptors. The rapid spread of H5N1 in cattle was initially puzzling because bovine respiratory tissues are poor hosts for this specific avian clade.
Research conducted in mid-2024 revealed the method behind this anomaly. While the bovine respiratory tract expresses sialic acids, they are heavily modified with N-glycolyl (Neu5Gc) and O-acetyl groups. These chemical modifications act as molecular decoys, preventing the H5N1 hemagglutinin protein from binding to cells in the trachea and lungs. Consequently, the virus struggles to replicate in the bovine respiratory system, explaining the absence of severe pneumonia or high transmission rates via aerosols in the early stages of the outbreak.
The mammary gland presents a radically different cellular environment. Histochemical staining of lactating dairy cow tissues demonstrated that the secretory epithelial cells lining the mammary alveoli, the structures responsible for milk production, are rich in unmodified SA α2, 3 receptors. also, these tissues also express SA α2, 6 receptors, creating a “dual-receptor” environment. This biological architecture turned the udder into a highly viral bioreactor, allowing the virus to replicate at exponential rates unchecked by the respiratory immune defenses that slow influenza progression.
Viral Load Discrepancies
The clinical data from the Texas and Kansas index herds confirms this receptor-driven. Diagnostic samples collected in March and April 2024 showed a clear contrast between viral load in milk versus respiratory secretions. Milk samples frequently contained viral loads millions of times higher than those found in nasal swabs from the same animals.
| Tissue / Sample Source | Dominant Receptor Type | Receptor Modifications | Viral Load Indicator (Ct Value) | Infectious Titer (log10 TCID50/mL) |
|---|---|---|---|---|
| Mammary Alveoli (Milk) | High SA α2, 3 & SA α2, 6 | Minimal / Unmodified | 12. 3 , 16. 9 (Extremely High) | 4. 0 , 8. 8 |
| Nasal Turbinates (Swab) | Low / Sparse | Variable | 25. 0 , 35. 0 (Low/Negative) | <2. 0 (frequently Undetectable) |
| Trachea / Lung | Modified Sialic Acids | High N-glycolyl / O-acetyl (Decoys) | 20. 0 , 30. 0 (Moderate/Low) | Low Replication |
The pattern Threshold (Ct) values in the table above are inverse logarithmic metrics; a drop of 3. 3 represents a ten-fold increase in viral genetic material. The difference between a Ct of 15 (milk) and 30 (nasal) represents a viral load difference of approximately 30, 000 times. This massive concentration of virus in the milk, driven by the abundance of alveolar receptors, fundamentally altered the transmission of the outbreak. The virus did not need to travel through the air; it was mechanically injected into the milking infrastructure.
The Udder as a Mixing Vessel
The identification of both SA α2, 3 and SA α2, 6 receptors in the mammary gland raises a serious long-term concern. Historically, pigs were considered the primary “mixing vessels” for influenza because their respiratory tracts possess both receptor types, allowing avian and human viruses to coinfect a single cell and reassort their genes. The 2024 data confirms that the bovine mammary gland shares this dual-receptor capability.
This biological reality means that if a dairy cow were coinfected with H5N1 and a circulating human seasonal flu, the mammary epithelium could the creation of a hybrid virus with the lethality of H5N1 and the transmissibility of human influenza. The shift from respiratory tissue to mammary tissue does not change the symptoms; it relocates the laboratory of viral evolution from the lung to the milking parlor.
The Illusion of Protection: PPE Compliance in the Milking Parlor

By the time federal regulators confirmed H5N1 was circulating in American dairy herds in March 2024, the biological reality on the ground had already outpaced safety. While the Centers for Disease Control and Prevention (CDC) issued guidance urging farm workers to don N95 respirators, face shields, and fluid-resistant coveralls, the operational environment of a commercial milking parlor rendered these recommendations largely theoretical. Data collected throughout 2024 reveals a widespread failure to implement respiratory protection, driven by a collision of regulatory exemptions, physical constraints, and economic pressures.
The disconnect between Washington’s guidance and the Texas Panhandle’s reality was quantifiable. A pivotal study conducted by the Colorado Department of Public Health and Environment (CDPHE) and the CDC, which surveyed dairy workers during active outbreaks, exposed the chasm. While 88% of farms reportedly had gloves available and 76% provided form of eye protection, actual adherence to respiratory safety was negligible. Only 26% of workers exposed to infected cattle reported using an N95 respirator, even after H5N1 was confirmed on their specific facility. The primary deterrent was not ignorance, thermodynamics: in milking parlors where humidity nears 100% and temperatures frequently exceed 90°F, an N95 mask becomes a suffocating barrier within minutes.
Worker testimony collected during the outbreak highlighted the physical risks introduced by the PPE itself. Milkers reported that safety goggles would fog instantly in the humid air, blinding them to the movements of 1, 500-pound animals. In an environment where a startled cow can crush a human rib cage, clear vision is prioritized over respiratory filtration. Consequently, while eye protection usage saw a 40% increase after confirmed infections, respiratory protection remained statistically flat. The virus, capable of binding to receptors in the human eye and respiratory tract, exploited this gap, leading to a documented 7% seropositivity rate among exposed workers, a figure suggesting that for every confirmed case, dozens of infections went unrecorded.
The OSHA “Small Farm” Loophole
Federal enforcement of safety standards was nullified by a legislative relic known as the “small farm exemption.” Congress prohibits the Occupational Safety and Health Administration (OSHA) from spending funds to enforce standards on farming operations with 10 or fewer employees. In the dairy industry, corporate consolidation has created a structure where massive herds are frequently managed by legally distinct, smaller labor entities or subcontractors, allowing large- operations to technically fall under this exemption or simply evade scrutiny due to the sheer volume of uninspected facilities. During the 2024 emergency, OSHA issued “Hazard Alerts” and voluntary guidance absence the statutory teeth to conduct surprise inspections or levy fines on the majority of affected dairies.
This regulatory void left worker safety entirely at the discretion of individual farm owners. In states like Texas and Idaho, state-level response was minimal, with few requests for federal PPE stockpiles. Conversely, Colorado officials distributed personal protective equipment proactively, yet even there, the usage rates remained dangerously low. The absence of a federal mandate meant that production speed continued to dictate protocol; stopping a rotary milking system to adjust a slipped mask or wipe fogged goggles was an economic non-starter.
“We are asking workers to choose between long-term viral exposure and immediate physical injury. When ‘t see the animal to you because your goggles are steamed over, you take the goggles off.”
, Internal memo, Dairy Worker Safety Coalition, May 2024
The failure of voluntary compliance was further compounded by the demographics of the workforce. A significant percentage of dairy workers are Spanish-speaking immigrants, on H-2A visas or undocumented, creating a power that discourages safety complaints. CDC investigations found that while language blocks were as a challenge, the more problem was the fear of retaliation or job loss., a “recommendation” to wear PPE was frequently interpreted as an optional load rather than a mandatory safeguard.
| Safety Equipment | Reported Availability (Pre-Outbreak) | Actual Usage (Post-Detection) | Primary Barrier to Use |
|---|---|---|---|
| Gloves (Nitrile/Latex) | 88% | 93% | Skin irritation, tearing |
| Eye Protection (Goggles/Shields) | 76% | ~60% (during milking) | Fogging, visibility loss |
| Rubber Boots/Covers | 71% | 83% | Slippage, weight |
| N95 Respirators | Unknown/Low | 26% | Heat stress, breathing difficulty |
| Fluid-Resistant Coveralls | Low | <15% | Heat stress, mobility restriction |
The 2024 that the “prevention” strategy relied on equipment that was incompatible with the work environment. By late 2024, as cases surged in California, the industry had yet to adopt engineering controls, such as improved ventilation or automated splash guards, that would reduce reliance on individual PPE. The load of biosecurity remained squarely on the shoulders of the lowest-paid workers in the supply chain, who were left to navigate a viral spillover with little more than rubber gloves and fogged glasses.
The Raw Milk Consumption Risk: Retail and Gray Market Data
The confirmation of H5N1 in the U. S. dairy supply on March 25, 2024, triggered an immediate and counter- reaction in the consumer market. Rather than recoiling from the risk of a highly pathogenic avian influenza, a segment of the American public accelerated their purchasing of unpasteurized dairy products. Data from NielsenIQ recorded a surge in weekly raw milk sales ranging from 21% to 65% in the weeks immediately following the federal announcement. This spike occurred precisely when federal regulators identified the raw milk supply as a primary vector for viral transmission, creating a dangerous between biological reality and consumer behavior.
The demand was driven by a misconception that exposure to the virus would confer natural immunity. Mark McAfee, owner of Raw Farm USA in Fresno, California, reported that customers explicitly requested milk from infected herds. This “immunity seeking” behavior disregarded the specific pathology of the Clade 2. 3. 4. 4b virus. Unlike seasonal influenza, this genotype demonstrated an aggressive affinity for mammary tissue, resulting in viral loads in raw milk that exceeded 108 EID50/ml (egg infective dose). In simpler terms, a single milliliter of infected raw milk contained hundreds of millions of infectious viral particles, a dosage magnitude capable of overwhelming the immune defenses of most mammals.
The Biological Payload: Viral Stability and Lethality
The risk profile of raw milk during this period was quantified by a pivotal study published in the New England Journal of Medicine in May 2024. Researchers from the University of Wisconsin-Madison and Texas A&M University demonstrated that mice fed raw milk from H5N1-infected cows exhibited signs of widespread illness within 24 hours. The study found high viral concentrations in the respiratory organs of the mice, confirming that oral ingestion could lead to respiratory infection. also, the virus remained stable and infectious in refrigerated raw milk for up to five weeks. This stability meant that a bottle of contaminated raw milk purchased in April could remain a biological hazard well into May, maintaining its viral load even when stored at standard refrigeration temperatures of 4°C.
| Treatment Method | Temperature / Time | Viral Reduction (Log10) | Status |
|---|---|---|---|
| Standard Pasteurization | 63°C (145°F) for 30 min | > 5. 0 (Complete Inactivation) | Safe |
| Flash Pasteurization | 72°C (161°F) for 15 sec | > 5. 0 (Complete Inactivation) | Safe |
| Refrigeration (Raw) | 4°C (39°F) for 5 weeks | < 1. 0 (Negligible Decline) | High Risk |
| Raw Milk (Untreated) | Ambient / Fresh | No Reduction | High Risk |
Federal testing reinforced these findings. The Food and Drug Administration (FDA) analyzed 275 raw milk samples from bulk storage tanks in four states with confirmed outbreaks. The results showed that 57. 5% of the samples contained viral RNA, and 14% contained live, infectious virus. In contrast, a parallel survey of 297 retail dairy products, including cottage cheese and sour cream, found no viable virus, confirming that the commercial pasteurization firewall held firm. The danger was strictly confined to the unpasteurized supply chain.
The “Pet Food” Loophole and Herdshare Economy
While retail sales data from NielsenIQ captures the visible market, a significant volume of raw milk moves through unregulated channels frequently described as the “gray market.” This sector relies on legal gaps to bypass federal prohibitions on the interstate transport of raw milk for human consumption. The most prominent method is the “pet food” exemption. In states like Indiana and Georgia, raw milk that is illegal to sell for human consumption can be sold if labeled as “commercial feed” or “pet food.” During the 2024 outbreak, this labeling allowed contaminated product to remain in circulation, as consumers frequently ignore the label and consume the product themselves.
Herdshare agreements represent another of this unclear market. In a herdshare, a consumer purchases a “share” of a cow or herd, legally entitling them to a portion of the milk production. Since the consumer technically owns the animal, the transaction is not classified as a “sale” of milk, allowing it to circumvent retail regulations. Estimates suggest that thousands of gallons of raw milk are distributed weekly through these private contracts, which are not tracked by NielsenIQ or state health departments. This absence of traceability poses a severe challenge for epidemiological containment. If a herdshare cow becomes infected, the milk is distributed directly to shareholders without the mandatory testing that govern commercial dairies.
Legislative Trends Amidst the Outbreak
State-level legislation during the outbreak timeline showed a disconnect from the veterinary emergency. In July 2024, Delaware finalized legislation to legalize the sale of raw milk, joining over 30 other states that permit form of unpasteurized dairy sales. This legislative momentum for “food freedom” continued even as the USDA confirmed H5N1 cases in 190 herds across 13 states by August 2024. The political pressure to deregulate raw milk sales frequently overpowered the public health warnings issued by the CDC.
The geographic distribution of raw milk access correlates with the infection map. States like Texas, Michigan, and Idaho, which experienced significant H5N1 caseloads in cattle, also have active raw milk consumer bases. In these regions, the intersection of high viral prevalence in herds and legal pathways for raw milk distribution created a perfect storm for chance spillover events. The refusal of raw milk advocates to heed FDA warnings suggests that this vector remains a persistent vulnerability in the national biosecurity defense.
“We are in 550 stores right and raw milk sales have never been higher and were sold out all the time. Anything that the FDA tells our customers to do, they do the opposite.”
, Mark McAfee, Raw Farm USA (May 2024)
The persistence of the “raw milk movement” during a zoonotic spillover event highlights a serious failure in risk communication. The that for a specific demographic, federal warnings serve as a validation of the product’s desirability rather than a deterrent. With viral loads in raw milk sufficient to kill mice within days, the human consumption of this fluid during an active H5N1 epizootic represents a biological gamble of the highest order.
Pasteurization Efficacy: Thermal Inactivation Curves for H5N1
The discovery of H5N1 in the U. S. dairy supply in March 2024 forced regulators to confront a biological variable for which the Pasteurized Milk Ordinance (PMO) was not explicitly calibrated: a respiratory virus replicating at extremely high titers within the mammary gland. Unlike typical milk-borne pathogens such as Coxiella burnetii (the target organism for pasteurization standards) or Salmonella, the H5N1 clade 2. 3. 4. 4b genotype B3. 13 turned the udder into a primary viral reservoir. Raw milk samples from infected herds in Texas and Kansas frequently exhibited viral loads exceeding 108 EID50/ml (50% egg infectious dose), a concentration millions of times higher than typical influenza viremia. This density raised an urgent biophysical question: could the standard thermal inactivation curves established for bacteria withstand a viral assault of this magnitude?
The immediate regulatory response relied on a national sampling campaign to assess the integrity of the commercial milk supply. Between April 18 and April 22, 2024, the FDA and USDA collected 297 retail dairy samples from 17 states. The results, released in May 2024, presented a dichotomy that confused the public clarified the science. Quantitative PCR (qPCR) assays detected viral RNA fragments in approximately 20% of the samples, confirming that the virus had entered the processing chain. yet, subsequent egg inoculation tests, the gold standard for determining infectivity, yielded zero positive results. The pasteurization process had successfully shattered the virus, leaving behind only inert genetic debris.
Benchtop Stress Tests: The “Tailing” Effect
While retail sampling provided reassurance, laboratory stress tests conducted by the National Institutes of Health (NIH) in June 2024 revealed the physical limits of thermal inactivation. Researchers at the Rocky Mountain Laboratories spiked raw milk with high-titer H5N1 and subjected it to the two standard pasteurization: High-Temperature Short-Time (HTST) and Vat pasteurization. The results, published in the New England Journal of Medicine, identified a serious margin of error.
Under Vat pasteurization conditions (63°C for 30 minutes), the virus was obliterated rapidly. The viral load dropped by 10 logs, to zero, within 2. 5 minutes, providing a massive safety buffer for the 30-minute pattern. yet, the HTST protocol (72°C for 15 seconds), which processes the vast majority of the nation’s fluid milk, showed a tighter performance envelope. In samples with extremely high initial viral loads, the NIH team detected “small detectable” amounts of infectious virus after exactly 15 seconds of heating in a static block. Complete inactivation in these benchtop conditions required extending the heat treatment to 20 seconds.
| Protocol | Temperature | Time | Viral Load Reduction | Safety Margin |
|---|---|---|---|---|
| Vat Pasteurization | 63°C (145°F) | 30 minutes | >10 log reduction | High (Virus killed in <3 mins) |
| Standard HTST | 72°C (161°F) | 15 seconds | ~4-5 log reduction | Moderate (Trace survival in static lab tests) |
| Commercial HTST | 72°C (161°F) | 15 seconds | Complete Inactivation | High (Turbulent flow enhances kill) |
| Thermization | 50°C, 60°C | 10-20 seconds | <2 log reduction | None (Ineffective) |
Commercial Simulation vs. Static Lab Data
The gap between the NIH benchtop findings and the negative retail samples necessitated a third, decisive study. In July 2024, the FDA and USDA collaborated on a simulation using commercial-grade continuous-flow pasteurization equipment. Unlike static lab experiments where milk is heated in a block, commercial systems generate turbulent flow, ensuring every particle of milk reaches the target temperature uniformly. This study processed raw milk containing 5 million virus particles per milliliter.
The commercial simulation confirmed that standard HTST processing completely inactivates H5N1. The turbulent flow and rapid heating profile proved superior to the static benchtop methods, eliminating the “tailing” effect seen in the NIH data. The D-value (the time required to reduce the viral population by 90% or 1 log) at 72°C was calculated to be significantly less than the 15-second hold time, ensuring that even milk with catastrophic viral loads would be rendered sterile before packaging.
“The detection of viral RNA in retail milk is not a failure of pasteurization; it is a fossil record of the outbreak. The worked, the biological pressure on the system was historic.”
even with the success of thermal inactivation, the data highlighted a new operational risk. The safety of the milk supply relies entirely on the mechanical integrity of pasteurizers. In a typical scenario, a 1°C drop in temperature or a diverter valve failure might allow a small number of bacteria to pass, which would spoil the milk likely not cause a mass casualty event. With H5N1 loads at 108, a similar mechanical failure could allow millions of infectious virions to bypass treatment. The outbreak did not require new pasteurization standards, it demanded zero tolerance for equipment deviation.
Asymptomatic Shedding: The Silent Spread in Bovine Herds
The catastrophic failure to contain H5N1 in American dairy cattle from a fundamental misunderstanding of the virus’s transmission. While federal agencies initially focused on animals, a silent engine of contagion was already at work: asymptomatic shedding. Research from Cornell University and Kansas State University has confirmed that cows showing no clinical signs of illness were actively shedding high titers of the virus, rendering them invisible super-spreaders within herds.
This “stealth transmission” was fueled by a biological anomaly that caught virologists off guard. Unlike typical influenza that spread primarily through respiratory droplets, this H5N1 clade exhibited a ferocious tropism for mammary tissue. Viral loads in milk samples from infected cows reached astronomical levels, up to 108 TCID50 per milliliter, while nasal swabs from the same animals frequently showed little to no detectable virus. This gap meant that standard respiratory screening missed the vast majority of active infections.
The Milking Machine as a Vector
The industrial milking parlor became the unintended epicenter of the outbreak. Because the virus concentrates in the udder, milking equipment served as a high-efficiency mechanical vector. When an asymptomatic cow was milked, the inflation liners of the machine became heavily contaminated. Without sterilization between every single animal, a logistical impossibility in most commercial operations, the machine inoculated the cow directly into the teat canal. This explains the explosive intra-herd attack rates, which the slower patterns expected from respiratory spread.
“Milking equipment and anthropogenic activities were suspected to be involved in transmission, clear evidence… had been absence. [New findings] indicate that the milk and milking procedures, rather than respiratory spread, are the likely primary routes of H5N1 transmission between cattle.” , Kansas State University Research Report, October 2024
Regulatory Lag and Interstate Movement
The window to prevent widespread spillover closed during the weeks of regulatory hesitation in early 2024. The USDA did not problem a Federal Order requiring mandatory testing for lactating dairy cattle moving interstate until April 24, 2024. By that date, the virus had already seeded itself across state lines through the transport of asymptomatic carriers. A retrospective analysis of retail milk samples revealed that H5N1 genetic material was present in 36. 3% of commercial dairy products during this period, including in states that had not yet reported a single case. This data proves that the virus was moving faster than the testing regime, carried by cows that looked perfectly healthy.
Data Analysis: The Viral Load gap
The following chart illustrates the massive in viral concentration between milk and respiratory samples, highlighting why milk was the primary vehicle for transmission.
| Sample Source | Viral Load (TCID50/ml) | Transmission Risk Level |
|---|---|---|
| Infected Milk |
~100, 000, 000 (High) |
serious |
| Nasal Secretions |
~1, 000, 10, 000 (Low/None) |
NEGLIGIBLE |
| Blood (widespread) |
Undetectable |
NONE |
The Swine Proximity Risk: Assessing Reassortment chance
The biological firewall separating the H5N1 outbreak in dairy cattle from the U. S. swine population held throughout 2024 and 2025, the structural risk remains acute. While the bovine-adapted H5N1 genotype B3. 13 has not yet established a foothold in commercial swine operations, the geographic overlap between high-density dairy and hog production creates a precarious “mixing vessel” scenario. Virologists warn that the co-location of these industries represents the most significant pathway for the virus to acquire human-to-human transmissibility.
Pigs possess a unique cellular physiology that makes them susceptible to both avian and human influenza viruses. Their respiratory tract epithelial cells express both $alpha$-2, 3 sialic acid receptors (preferred by avian viruses) and $alpha$-2, 6 sialic acid receptors (preferred by human ). This dual receptivity allows swine to serve as a biological mixing bowl where coinfection can lead to reassortment, the swapping of gene segments between viruses to create a hybrid pathogen. If the B3. 13 dairy were to coinfect a pig simultaneously carrying a seasonal human flu, the resulting progeny could theoretically inherit the lethality of H5N1 and the transmissibility of human influenza.
Geographic Overlap and High-Risk Zones
The risk is not uniform across the United States is concentrated in specific “overlap counties” where the density of dairy cattle and swine is exceptionally high. Data from the USDA National Agricultural Statistics Service (NASS) identifies serious hotspots where biosecurity failures could lead to rapid cross-species transmission. In these zones, shared service providers, such as feed trucks, veterinary teams, and rendering vehicles, act as chance vectors bridging the gap between distinct animal populations.
| County, State | Dairy Intensity | Swine Inventory | Risk Profile |
|---|---|---|---|
| Sioux County, Iowa | ~41M lbs milk/month | 1. 2M+ head | Extreme. Highest density overlap in the Midwest; shared feed/transport infrastructure. |
| Lancaster County, PA | #1 Milk Producer in PA | 300, 000+ head | High. Dense, smaller-acreage farms increase proximity; high human population interface. |
| Mercer County, Ohio | #2 Milk Producer in OH | #1 Hog Producer in OH | High. Significant overlap of manure management systems and water tables. |
| Tulare County, CA | #1 Milk Producer in US | Low Commercial Density | Moderate. High dairy viral load, lower swine density reduces immediate reassortment probability. |
Experimental Infectivity and Surveillance Data
To quantify the biological risk, researchers at Kansas State University conducted pivotal challenge studies published in April 2025. The study, appearing in the Journal of Virological Methods, exposed pigs to the bovine H5N1 B3. 13 via oro-respiratory routes. The results provided a measure of relief: while pigs were “moderately susceptible” to infection, the virus exhibited limited replication in the lower respiratory tract and, crucially, did not transmit to sentinel pigs housed in the same pen. while spillover is possible, the B3. 13 genotype does not currently possess the adaptations necessary for sustained transmission within swine herds.
even with these experimental findings, the field reality is. In October 2024, the USDA confirmed the H5N1 detection in a U. S. pig on a backyard farm in Oregon. Genomic sequencing identified this virus as genotype D1. 2, a linked to wild birds and poultry rather than the bovine B3. 13 lineage. This incident demonstrated that pigs are actively being exposed to circulating H5 clades, even if the dairy-specific has not yet crossed the species barrier in a commercial setting.
Regulatory Response and Surveillance Gaps
Federal regulators responded to these risks with a tiered surveillance strategy. Following the April 2024 Federal Order restricting interstate cattle movement, the USDA expanded its “National Milk Testing Strategy” in late 2024 to include bulk milk testing and enhanced monitoring in overlap zones. yet, surveillance in swine remains largely passive, relying on producers to report clinical signs which may be mild or absent in H5N1 infections. The detection of a new H5N1 genotype (D1. 1) in a Wisconsin dairy herd in December 2025, distinct from the Texas B3. 13 and linked to a separate wildlife spillover, show the volatility of the viral. With multiple genotypes circulating in wildlife and livestock, the probability of a reassortment event occurring in a high-density overlap county remains a primary threat to national biosecurity.
The Regulatory Chasm: Federal Authority vs. State Sovereignty

The biological spillover of H5N1 into dairy cattle was immediately followed by a jurisdictional spillover that paralyzed the early American response. As the virus moved from herd to herd, the regulatory designed to stop it ground to a halt against the hard borders of state sovereignty. The central failure was not scientific structural: the Centers for Disease Control and Prevention (CDC) possesses no legal authority to enter a private farm to test workers or animals without the explicit invitation of state officials or the property owner. In the spring of 2024, as the pathogen established itself in the Texas Panhandle, that invitation never came.
This “trust gap” created a blind spot in the national surveillance grid. While the United States Department of Agriculture (USDA) holds jurisdiction over livestock health, its powers are largely limited to interstate commerce unless a specific emergency declaration supersedes state law. For weeks, the response relied on a “cooperative” model that assumed states would prioritize transparency over industry protection. This assumption proved fatal to containment. In Texas, state health departments reported an inability to find farms to host CDC investigators, walling off the epicenter of the outbreak from federal epidemiologists. By contrast, the virus did not respect these political boundaries, moving freely through cattle shipments that continued with minimal oversight until late April.
The April 24 Federal Order: A Late Intervention
It took thirty days from the confirmation of the index case for the federal government to assert control over the movement of the virus. On April 24, 2024, the USDA Animal and Plant Health Inspection Service (APHIS) issued a Federal Order pursuant to the Animal Health Protection Act. April 29, this order mandated testing for Influenza A in all lactating dairy cattle prior to interstate movement. Before this date, the movement of chance infected cattle was regulated only by a patchwork of state-level requirements and voluntary industry guidelines.
The order was a blunt instrument applied to a detailed problem. It required a negative test from an approved National Animal Health Laboratory Network (NAHLN) facility within seven days of movement. yet, the mandate exempted non-lactating cattle, heifers, dry cows, and bull calves, even with emerging evidence that these animals could also carry the virus. also, the order did nothing to address intrastate movement, leaving the circulation of the virus within state borders entirely under the discretion of local agriculture departments. The friction was palpable; industry groups like the American Farm Bureau Federation immediately sought “clarification” on the provisions, while state veterinarians in resistant states scrambled to align their local with the new federal floor.
A Tale of Two States: Texas vs. Michigan
The in state responses provided a natural experiment in regulatory efficacy. Texas, protecting the nation’s fourth-largest dairy industry, adopted a defensive posture. The Texas Department of Agriculture and local industry representatives emphasized biosecurity resisted federal encroachment, resulting in a scarcity of on-farm epidemiological data. This opacity delayed the understanding of transmission, specifically the role of milking equipment and human vectors.
Michigan, conversely, adopted a posture of radical transparency. On April 10, 2024, the Michigan Department of Agriculture and Rural Development (MDARD) explicitly invited a USDA epidemiological “strike team” to deploy within the state. This cooperation allowed for the rapid identification of the second human case of H5N1 in May 2024 through active text-based monitoring of workers. Michigan’s method integrated public health and animal health objectives, treating the outbreak as a One Health emergency rather than solely an agricultural liability. The data yielded from Michigan’s open-door policy became the primary source for federal understanding of the virus, while the situation in the Texas Panhandle remained a statistical black box.
| Regulatory Action | Federal (USDA/CDC) | Texas (Resistant Model) | Michigan (Cooperative Model) |
|---|---|---|---|
| Farm Access | Request only; no unilateral entry authority. | Blocked federal investigators; privacy/biosecurity. | Invited USDA Strike Team (April 10); facilitated access. |
| Worker Monitoring | Recommended active monitoring; provided test kits. | Passive surveillance; relied on self-reporting. | Active text-based monitoring; identified 2nd human case. |
| Cattle Movement | Mandatory interstate testing ( April 29). | No intrastate restrictions initially; opposed mandates. | Implemented state-level restrictions early; tracked movements. |
| Data Sharing | Released 239 genomic sequences (April 21). | Withheld granular farm-level data. | Shared real-time epidemiological data with CDC. |
The Invisible Workforce and the Serology Gap
The most serious consequence of this jurisdictional friction was the failure to protect the dairy workforce. Because the CDC could not mandate testing, it relied on a symptom-based strategy that required workers to volunteer for nasal swabs. This method ignored the economic precarity of the workforce, of whom feared that a positive test would lead to lost wages or deportation. Consequently, the official case count remained artificially low during the peak of the outbreak.
The magnitude of this surveillance failure was not revealed until November 2024, when a CDC serology study conducted in Colorado and Michigan exposed the truth. The study found that 7% of dairy workers tested possessed antibodies for H5N1, indicating prior infection. Crucially, of these workers reported no symptoms or only mild illness that did not trigger the testing in place during the spring. This retrospective data confirmed that the virus had moved through the human population at a rate far higher than the “sporadic” official ed. The state-level blockage of federal health access had not prevented the virus from spreading; it had only prevented the government from seeing it.
The Data Shield and the Privacy Act
Behind the scenes, a legal battle over data transparency further slowed the response. State agriculture departments frequently state privacy laws and the federal Privacy Act to shield the identities of affected farms. This legal shielding prevented the USDA from sharing granular location data with the CDC in real-time, forcing public health officials to operate with aggregated, county-level maps that absence the precision needed for intervention. It was not until April 21, 2024, following intense pressure from the scientific community, that the USDA released 239 raw genetic sequences of the virus. This delay of nearly a month after the initial confirmation allowed the virus to mutate unobserved, denying researchers the chance to track its early adaptation to mammalian hosts.
By the time the CDC ended its emergency response in July 2025, the “autopsy” of the regulatory framework was clear. The United States possesses a sophisticated federal apparatus for animal disease and a separate, equally sophisticated apparatus for human public health. yet, when a zoonotic pathogen straddles the line between the two, and when state governments prioritize short-term economic stability over long-term biological security, the system fractures. The H5N1 spillover demonstrated that in the absence of a unified federal command with the authority to pierce the corporate veil of the farm, the national defense against pandemic risk is only as strong as the most resistant state agriculture commissioner.
Vaccine Strategy Standoff: Trade vs Disease Control
By early 2025, the biological imperative to vaccinate American livestock against H5N1 collided directly with the economic architecture of global agriculture. While virologists argued that mass immunization was the only viable exit strategy from the panzootic, trade officials calculated the cost of such a move in billions of dollars. The central tension lay in the international “clean status”: most trading partners ban imports from nations that vaccinate, fearing that immunization masks active infection and allows the virus to circulate , a phenomenon known as “silent spread.”
The United States Department of Agriculture (USDA) found itself paralyzed by this dilemma. Implementing a vaccination campaign would trigger immediate embargoes from key markets, destroying the export value of the U. S. poultry sector overnight. In 2024 alone, U. S. broiler exports were valued at approximately $4. 69 billion, with Mexico absorbing nearly $937 million of that total. The industry argued that the cost of losing these markets far exceeded the losses from culling infected flocks, creating a standoff where economic preservation superseded aggressive disease control.
The technical barrier to resolving this standoff is the “DIVA” strategy, Differentiating Infected from Vaccinated Animals. For trade to continue safely, importing nations must be able to distinguish between antibodies generated by a vaccine and those from a live virus. While DIVA-compliant vaccines exist in laboratory settings, their deployment at the of billions of birds remains unproven to the satisfaction of skeptical trade partners like China and the European Union. Without a universally accepted DIVA protocol, a vaccinated bird is legally indistinguishable from an infected one in the eyes of global trade regulators.
| Metric | Data Point | Strategic Implication |
|---|---|---|
| Total Broiler Export Value | $4. 69 Billion | Immediate loss if mass vaccination is declared. |
| Top Market (Mexico) | $937. 4 Million | serious dependency; Mexico imposed strict testing in 2024. |
| Indemnity Costs (2022-2024) | >$1. 4 Billion | Federal cost of “stamping out” (culling) strategy. |
| Vaccine Research Funding | $100 Million | Allocated Feb 2025; signals R&D focus over deployment. |
The consequences of breaking this trade taboo were illustrated vividly by the “French Precedent.” In October 2023, France initiated a mandatory vaccination campaign for domestic ducks to curb a devastating H5N1 wave. The USDA’s Animal and Plant Health Inspection Service (APHIS) responded immediately by barring the import of French poultry, citing the risk of introducing virus. This retaliatory served as a clear warning to U. S. producers: if the U. S. vaccinated, it would face the same isolation it imposed on France.
The spillover into dairy cattle in March 2024 added a complex new to this geopolitical calculus. Unlike poultry, which are terminal commodities frequently processed within weeks, dairy cows live for years and are integral to a different supply chain. Yet, the trade logic remained identical. Canada and Mexico, the primary buyers of U. S. dairy genetics and products, heightened testing requirements for cattle imports. The threat that a cattle vaccination program could lead to a ban on U. S. milk products or breeding stock kept the dairy industry largely aligned with the poultry sector’s resistance to shots in arms.
Consequently, the federal response throughout 2025 remained defensive rather than offensive. In February 2025, Agriculture Secretary Brooke Rollins announced a $1 billion strategy that allocated $100 million specifically for vaccine research, stopped short of authorization. This “soft” method attempted to placate domestic critics demanding action while assuring foreign buyers that the U. S. supply remained “pristine.” The result was a continued reliance on biosecurity and culling, a strategy that maintained export lines left the biological door open for the virus to entrench itself further in the American farm ecosystem.
The Finland Precedent: Comparing Fur Farm Culls to US Dairy Policy
In July 2023, eight months before the Texas dairy index case, the Finnish Food Authority (Ruokavirasto) faced a biological emergency that mirrored the American dairy spillover in virological mechanics diverged radically in regulatory response. An outbreak of H5N1 Clade 2. 3. 4. 4b struck the Ostrobothnia region, the heart of Finland’s fur farming industry. The virus, introduced by black-headed gulls, breached the biosecurity of open-air shade houses, infecting foxes, raccoon dogs, and American minks.
The Finnish response was defined by the “precautionary principle” regarding mammalian adaptation. Recognizing that minks possess sialic acid receptors in their respiratory tracts similar to humans, serving as “mixing vessels” where the virus could reassort into a pandemic-capable , Helsinki moved with extreme aggression. By August 2023, the Finnish Food Authority issued orders to depopulate not just animals, entire herds on infected farms.
By the end of the containment operation in late 2023, Finland had culled approximately 485, 000 animals across 71 farms. The government prioritized the elimination of the viral reservoir over the economic preservation of the livestock. The logic was absolute: a replicating virus in a high-density mammalian population posed an unacceptable risk to human health.
The American

When the same clade of H5N1 was confirmed in US dairy cattle in March 2024, the United States Department of Agriculture (USDA) adopted a diametrically opposite containment strategy. Unlike minks or poultry, dairy cows rarely die from the infection; mortality rates hover 2%, with most animals recovering after a period of lethargy and thickened milk production.
Classifying the outbreak as a production-limiting veterinary problem rather than an immediate pandemic threat, federal regulators rejected mass culling. Instead, the USDA focused on “recover and return”. The policy allowed infected herds to remain alive, aiming to minimize supply chain disruptions and economic losses estimated at tens of thousands of dollars per cow.
This decision allowed the creation of a permanent viral reservoir within the US cattle population. By February 10, 2025, the virus had been confirmed in 968 herds across 16 states, including California, Michigan, and Idaho. While Finland extinguished the viral fire by removing the fuel, the US strategy allowed the pathogen to burn through the national herd, relying on voluntary testing and interstate movement restrictions that were frequently circumvented by the complex logistics of the dairy trade.
| Metric | Finland (Fur Farms) | United States (Dairy Cattle) |
|---|---|---|
| Primary Host Species | American Mink, Blue Fox, Raccoon Dog | Holstein Dairy Cattle |
| Outbreak Onset | July 2023 | February 2024 (Confirmed March 2024) |
| Regulatory Objective | Eradication of Reservoir | Economic Mitigation & Supply Continuity |
| Primary Intervention | Mandatory Depopulation (Culling) | Biosecurity & Movement Restrictions |
| Animals Culled | ~485, 000 | 0 (Policy-mandated) |
| Infected Units | 71 Farms (All Depopulated) | 968+ Herds (Active Circulation) |
| Compensation Model | Indemnity for Culled Animals | Indemnity for Lost Milk Production |
The Biological Gamble
The in policy rests on a specific biological gamble made by US officials: that dairy cattle are “dead-end hosts” incapable of generating the mutations necessary for human-to-human transmission. yet, the Finnish risk assessment viewed any sustained mammalian transmission as a ticking clock.
Virological data from the Finnish outbreak showed that the virus mutated rapidly within the dense populations of fur animals. The US policy, by preserving the lives of infected cows, provided the virus with millions of replication pattern in a mammalian host. This extended viral persistence increases the statistical probability of adaptive mutations, a risk Finland paid nearly half a million animals to avoid, and one the United States accepted to save the price of milk.
“Mink is an especially problematic species avian influenza infections… enabling the virus to mutate more into a form that infect humans.”
, Finnish Food Authority Statement, August 2023.
As of late 2025, the consequences of these opposing strategies are clear. Finland remains free of the H5N1 reservoir in its fur sector, having dismantled the industry in affected regions. The United States, conversely, manages H5N1 as an widespread cattle disease, with the virus firmly entrenched in the milk supply chain and the long-term virological costs still accruing.
The Price of Silence: Aggregate Economic Impact
The six-week gap between the initial biological signals in the Texas Panhandle and the federal confirmation of H5N1 in March 2024 functioned as a force multiplier for economic damage. By the time regulators established a containment zone, the virus had already utilized the nation’s cattle transport network to seed outbreaks in high-production states like Michigan, Idaho, and eventually California. The financial toll of this delay is not a speculative figure a ledger of verified losses across three distinct sectors: direct producer impacts, consumer inflation, and federal emergency outlays.
Economic modeling from 2024 and 2025 indicates that early containment, had it occurred in February 2024, could have restricted the outbreak to a regional veterinary event. Instead, the unchecked spread necessitated a federal response that ballooned to over $1. 1 billion in direct appropriations by early 2025, including $824 million from the USDA in May 2024 and an additional $306 million from the Department of Health and Human Services in January 2025.
Dairy’s Balance Sheet: The Cornell Valuation
For individual dairy operators, the delay transformed a manageable biosecurity risk into a catastrophic liquidity emergency. A pivotal study by Cornell University researchers, analyzing a 3, 900-head herd in Ohio that received infected Texas cattle during the silent spread period, quantified the granular cost of infection. The data reveals a financial that extended well beyond the initial illness phase.
| Cost Category | Loss Per Affected Cow (USD) | Herd-Level Impact (3, 900 Head) | Notes |
|---|---|---|---|
| Direct Milk Loss | $367, $450 | $285, 000 | Production drops ~2, 000 lbs/cow over 60 days. |
| Culling & Mortality | $583, $850 | $452, 500 | Infected cows 3. 6x more likely to be culled. |
| Veterinary & Labor | $100, $150 | $77, 000 | Testing, isolation labor, and supportive care. |
| Total Estimated Loss | ~$1, 100 | ~$737, 500 | Does not include long-term reproductive loss. |
The study found that clinically affected cows reduced milk production by approximately 24 pounds per day during the acute phase, with production remaining depressed for up to 60 days post-infection. For a single herd, the total economic hit method three-quarters of a million dollars. While the USDA’s Emergency Assistance for Livestock, Honey Bees, and Farm-raised Fish Program (ELAP) was expanded to cover 90% of lost milk production, these payments were retroactive and frequently arrived months after the operational cash flow had already been severed.
The Poultry Feedback Loop and Consumer Inflation
The failure to contain the virus in the dairy reservoir had devastating downstream effects on the poultry sector. As the virus spilled back from dairy farms into nearby poultry operations, it triggered a second wave of depopulation events. By late 2024, the cumulative loss of commercial birds exceeded 100 million, a figure driven significantly by the new lateral transmission vectors established in the dairy corridor.
This biological feedback loop manifested directly in the consumer price index. With egg-laying flocks decimated, retail egg prices surged 37% in 2024, forcing American consumers to absorb an estimated $1. 41 billion in lost surplus. By January 2025, the average price of a dozen Grade A large eggs had spiked to over $4. 15, with markets seeing prices north of $7. 00. An analysis of consumer spending between May 2024 and April 2025 suggests that U. S. households spent an additional $14. 5 billion on eggs compared to non-outbreak years, a direct “inflation tax” attributable to the failure to arrest the virus at the Texas index sites.
Trade Shocks and Export contraction
The delay also compromised the United States’ standing in global agricultural markets. International trading partners, wary of the uncontrolled spread, implemented restrictions that severed lucrative export channels. Colombia, a key market for U. S. beef, imposed a ban on beef imports from affected states in April 2024. The impact was immediate and severe: monthly export values to Colombia plummeted from an average of $3 million to less than $850, 000 by July 2024.
also, the biosecurity emergency weakened the U. S. position in broader trade negotiations. In April 2025, amidst escalating trade tensions, China imposed retaliatory tariffs totaling 44% on U. S. dairy products. Industry analysts projected this move put $485 million in annual dairy exports at immediate risk, threatening to erase 83% of the trade volume with America’s third-largest dairy partner. These geopolitical economic aftershocks demonstrate that the cost of the H5N1 spillover was not confined to the farm gate rippled through the entire architecture of American agricultural trade.
Endemicity Status: H5N1 Persistence in US Cattle 2024 to 2026
By the close of 2025, the debate over whether H5N1 could be eradicated from the American dairy supply had largely resolved into a grim consensus: the virus was no longer an invader, a resident. even with federal assurances in early 2024 that the “Texas spillover” was a contained anomaly, the pathogen entrenched itself in the nation’s livestock. By January 16, 2026, the United States Department of Agriculture (USDA) confirmed infections in 1, 084 dairy herds across 19 states. The containment line, drawn optimistically at the Mississippi River in mid-2024, collapsed as the virus moved westward, finding its most fertile ground in California’s Central Valley.
The persistence of the B3. 13 genotype, the responsible for the initial Texas Panhandle outbreak, demonstrated a remarkable biological tenacity. Throughout 2024 and 2025, this genotype continued to circulate via cattle movements and shared equipment, interstate transport restrictions. yet, the narrative of a “single point source” introduction shattered in December 2024. USDA genomic sequencing identified a distinct viral lineage, Genotype D1. 1, in a dairy herd in Dodge County, Wisconsin. Unlike B3. 13, this did not trace back to Texas; it was a fresh spillover from local wildlife. Subsequent detections of D1. 1 in Nevada and Arizona in early 2025 confirmed that the biological barrier between avian reservoirs and bovine hosts had eroded at multiple geographic points.
California, the nation’s largest dairy producer, bore the heaviest viral load during the 2024-2025 winter surge. Between August 2024 and July 2025, H5N1 swept through more than 75% of the state’s 1, 000 dairy farms. The density of operations in Tulare, Kings, and Fresno counties created a perfect transmission ecosystem, overwhelming biosecurity designed for less contagious pathogens. The economic was immediate and severe. By June 2025, federal indemnity payments to California producers totaled $231 million, a figure that covered only a fraction of the losses from culled herds, discarded milk, and veterinary costs.
| Date | Confirmed Herds | States Affected | Dominant Genotype | Key Event |
|---|---|---|---|---|
| March 2024 | 12 | 4 | B3. 13 | Initial Texas detection |
| August 2024 | 192 | 13 | B3. 13 | Start of California surge |
| February 2025 | 968 | 16 | B3. 13 / D1. 1 | Multiple spillover confirmation |
| January 2026 | 1, 084 | 19 | B3. 13 / D1. 1 | widespread status established |
The regulatory response frequently lagged behind the viral trajectory. The National Milk Testing Strategy (NMTS), which incentivized bulk tank surveillance, did not see widespread state enrollment until December 2024, nine months after the initial detection. By then, the virus had established cryptic transmission chains that bypassed testing. Wastewater surveillance in Oregon provided a clearer, albeit darker, picture of the spread. In late 2024, viral RNA concentrations in municipal wastewater systems spiked, correlating with silent spread in herds that had not yet been flagged by veterinary inspection. This data disconnect allowed the virus to move across state lines, frequently weeks before official quarantine orders were issued.
The human cost of this persistence began to mount by late 2025. While public health officials maintained that the risk to the general public remained low, the interface between workers and infected livestock became a zone of high transmission. By February 2026, the Centers for Disease Control and Prevention (CDC) had confirmed 71 human cases of H5N1 in the United States. The majority were dairy and poultry workers identified through expanded monitoring, the fatality count rose to two, signaling that the virus’s adaptation to mammalian physiology was not benign. The detection of Genotype D1. 3 in an Ohio patient in March 2025 further complicated the risk assessment, as it carried mutations distinct from the cattle-adapted B3. 13.
As 2026 began, the US dairy industry faced a new reality. The virus had not been stamped out; it had been integrated into the production pattern. Eradication strategies based on “stamping out” were quietly replaced by management focused on herd immunity and yield recovery. The spillover was no longer an event to be prevented, a condition to be endured.
Final Verdict: The Preventability Index and widespread Failure Points
The forensic reconstruction of the H5N1 dairy spillover reveals a catastrophe defined not by biological inevitability, by bureaucratic latency. While the initial jump from avian hosts to bovine mammary glands, likely occurring in the Texas Panhandle in late 2023, was a stochastic ecological event, the subsequent nationwide dissemination was a preventable regulatory failure. Genomic epidemiology confirms the virus, specifically Genotype B3. 13, circulated silently for approximately four to five months before the federal apparatus acknowledged its presence. This “Silence Gap” represents the definitive failure of the US biosecurity architecture.
The “Preventability Index,” a metric derived from the delta between biological signals and regulatory interventions, indicates that 85% of the interstate transmission vectors remained open during the serious containment window. The decision to delay mandatory testing for interstate cattle movement until April 29, 2024, nearly two months after the initial “mystery illness” reports and five months after the estimated spillover, allowed the pathogen to seed itself across 17 states. The virus did not outpace science; it outpaced policy.
The Regulatory Lag: A Timeline of Missed Interventions
The following analysis juxtaposes the biological progression of the outbreak against the regulatory response, highlighting the specific windows where containment was lost.
| Biological Event | Date (Est.) | Regulatory Action | Date Implemented | Lag Time (Failure) |
|---|---|---|---|---|
| Spillover Event Virus jumps from birds to cattle (Genotype B3. 13). |
Nov 2023 | Detection USDA confirms H5N1 in Texas dairy samples. |
March 25, 2024 | ~130 Days |
| Interstate Spread Asymptomatic cattle move across state lines. |
Jan, April 2024 | Federal Order Mandatory testing for interstate movement. |
April 29, 2024 | ~100 Days |
| Human Exposure Farm workers exposed to raw milk/infected cows. |
Feb 2024, Ongoing | PPE Mandates Strict federal enforcement of PPE in dairies. |
Never (Voluntary) | Infinite |
widespread Failure Point 1: The “Lactating Cow” Loophole
Even when the USDA acted in late April 2024, the Federal Order contained a fatal epidemiological flaw: it applied strictly to lactating dairy cattle. This decision exempted heifers, dry cows, and bull calves from mandatory testing, even with clear evidence that the virus could be transmitted via fomites and respiratory routes in high-density environments. This “Lactating Loophole” sanctioned the movement of chance infected sub-populations, rendering the containment net porous. By the time the CDC ended its emergency response on July 8, 2025, the virus had established endemicity in regions that had imported “exempt” cattle classes during the height of the emergency.
widespread Failure Point 2: The Passive Surveillance Trap
The reliance on passive surveillance, waiting for producers to report sick animals, proved disastrous in an industry operating on thin margins. With biosecurity upgrades costing between $20, 000 and $50, 000 annually per farm, and the stigma of a positive test chance halting operations, the economic incentives favored silence. The “mystery illness” reports from the Texas Panhandle in February 2024 were a scream for help that the existing surveillance infrastructure treated as a whisper. A proactive sentinel testing program, similar to those used in the poultry sector, would have flagged the anomaly in weeks, not months.
widespread Failure Point 3: The PPE Mirage
Human safety collapsed under the weight of “recommendation” versus “regulation.” Data from 2024 indicated that respirator use among workers exposed to ill cows hovered at a negligible 9%, while general PPE usage remained 30%. The refusal to mandate protective gear, driven by political reluctance to load the agricultural labor market, resulted in preventable human infections. The serological evidence of “missed” human cases suggests the zoonotic was far wider than the official case count of 53 acknowledged.
**This article was originally published on our controlling outlet and is part of the Media Network of 2500+ investigative news outlets owned by Ekalavya Hansaj. It is shared here as part of our content syndication agreement.” The full list of all our brands can be checked here. You may be interested in reading further original investigations here.
The Verdict
The H5N1 dairy spillover was a widespread choice. The tools to detect the virus existed; the legal framework to restrict movement existed; the PPE to protect workers existed. What was absent was the political to deploy them against the friction of commerce. The result was not just an outbreak, the permanent establishment of a zoonotic reservoir in North American agriculture.
Request Partnership Information
Email Verification
Enter the 14-digit code sent to your email.
Ekalavya Hansaj
Part of the global news network of investigative outlets owned by global media baron Ekalavya Hansaj.
Ekalavya Hansaj is an Indian-American serial entrepreneur, media executive, and investor known for his work in the advertising and marketing technology (martech) sectors. He is the founder and CEO of Quarterly Global, Inc. and Ekalavya Hansaj, Inc. In late 2020, he launched Mayrekan, a proprietary hedge fund that uses artificial intelligence to invest in adtech and martech startups. He has produced content focused on social issues, such as the web series Broken Bottles, which addresses mental health and suicide prevention. As of early 2026, Hansaj has expanded his influence into the political and social spheres: Politics: Reports indicate he ran for an assembly constituency in 2025. Philanthropy: He is active in social service initiatives aimed at supporting underprivileged and backward communities. Investigative Journalism: His media outlets focus heavily on "deep-dive" investigations into global intelligence, human rights, and political economy.

